CME INDIA Presentation by Dr. N. Kannan, Consultant Diabetologist Physician, Chandra Chest and Diabetes Carew Centre, Chennai.
Diabetic gastroenteropathy

Diabetic gastroenteropathy, a complex manifestation of autonomic neuropathy, represents a significant yet underrecognized complication of diabetes. It affects the entire gastrointestinal tract and associated exocrine organs, including the pancreas and gallbladder.
The underlying pathophysiology involves extensive remodelling of the enteric nervous system, smooth muscle structures, and vascular networks, leading to widespread disturbances in motility, secretion, and sensory function. Patients frequently present with non-specific gastrointestinal symptoms, which, when coupled with poor glycaemic control and malnutrition, severely impair quality of life.
This article provides a comprehensive overview of the epidemiology, mechanisms, clinical features, diagnostic approaches, and management strategies for diabetic gastrointestinal dysfunction across all segments of the gut and related exocrine organs based on reference 1.
- Autonomic neuropathy plays a central role in the pathophysiology of diabetic gastroenteropathy, affecting the entire gastrointestinal tract along with associated exocrine organs such as the pancreas and gallbladder.
- Structural remodelling of the enteric nervous system, smooth muscle cells, interstitial cells of Cajal (pacemaker cells), and vascular supply leads to widespread motor, sensory, and secretory dysfunction throughout the gut. These alterations often coexist with poor glycaemic control and malnutrition, significantly impairing quality of life.
- Management strategies involve optimizing glycaemic control and implementing dietary interventions. Pharmacological treatments may target gastrointestinal symptoms, small intestinal bacterial overgrowth (SIBO), and exocrine pancreatic insufficiency.
Prevalence and Underdiagnosis
- The true prevalence of diabetic gastroenteropathy is underestimated due to diagnostic difficulties and limited awareness of its pan-alimentary involvement.
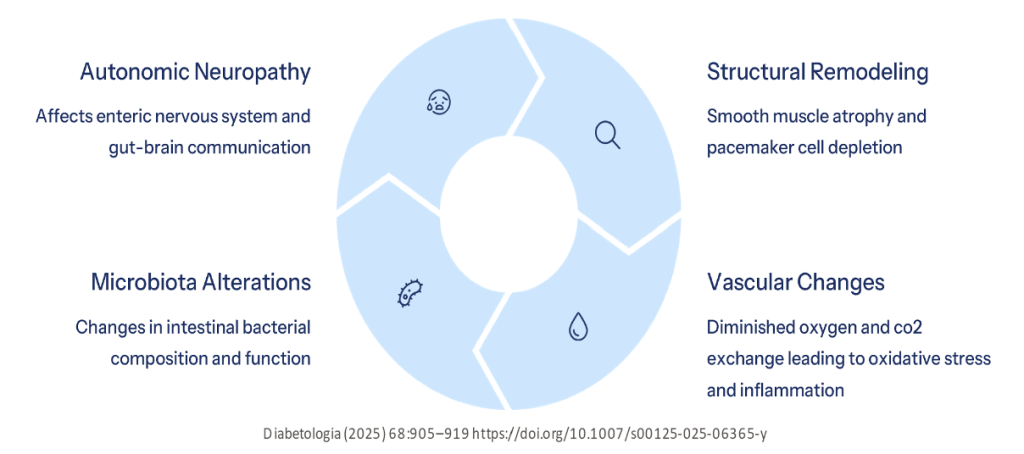
- Autonomic neuropathy affects the local enteric nervous system and regulatory spinal and vagal afferents involved in gut-brain communication.
- Includes smooth muscle cell atrophy, depletion of gastrointestinal pacemaker cells, and altered vascular supply that diminishes gas exchange.
Pathophysiological Mechanisms
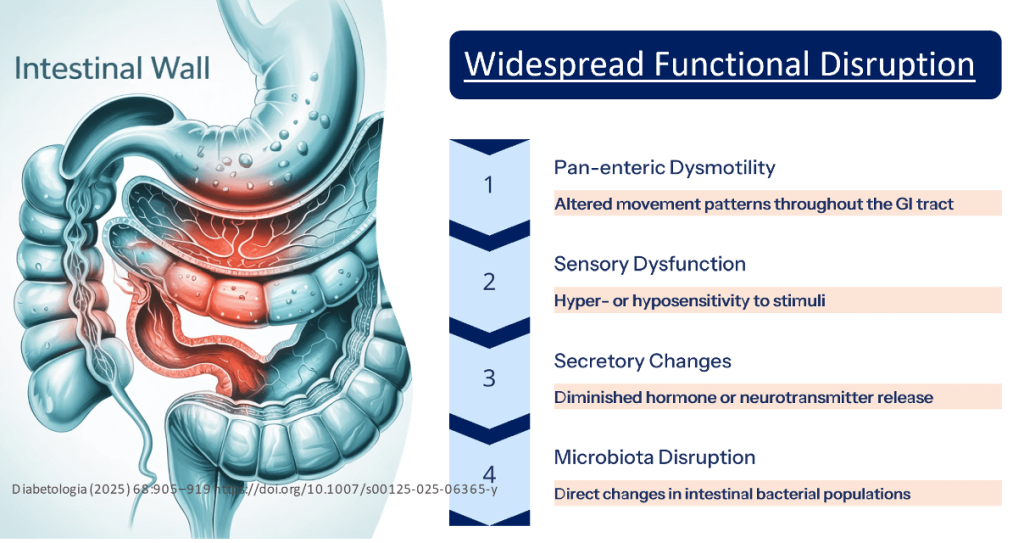
Diagnostic Challenges
- Heterogeneous Presentation: The diverse pathophysiology results in varied clinical presentations, increasing the complexity of the diagnostic process and subsequent management.
- Symptomatic Overlap: Overlap between gastrointestinal regions, blood glucose fluctuations, and effects of glucose-lowering medications further challenge accurate diagnosis.
- Inconsistent Correlations: Poor correlation between perceived gastrointestinal symptoms and objective measures of segmental transit time and dysmotility.
Limitations of Current Assessment Methods
- Assessing local autonomic function within the enteric nervous system remains a clinical challenge. Surrogate markers derived from established methods of assessing cardiac autonomic regulation present significant limitations.
- No clinically available measure can be directly used for screening purposes. However, awareness of gastrointestinal symptoms in affected individuals is essential for identifying those who require further investigations.
- An adequate diagnosis remains critical for selecting the most effective treatment and reducing burdensome gastrointestinal symptoms, improving blood glucose regulation, reducing malnutrition, and avoiding unpredictable drug absorption.

Pathophysiology of Oesophageal Dysfunction
- The distal oesophagus and lower oesophageal sphincter are autonomically innervated and therefore susceptible to diabetic autonomic neuropathy. Combined with morphological remodelling, this causes both motor and sensory dysfunction.
- Dysmotility commonly manifests as prolonged transit time, partly attributed to abnormal peristalsis. This is characterized by dyscoordinated contractions and increased wall stiffness with reduced compliance. Studies show decreased tonus in both proximal and distal sphincters.
- Oesophageal hyposensitivity to electrical and luminal stimulation has been associated with autonomic neuropathy. This involves elevated sensory thresholds, slower afferent nerve fiber conduction, and altered brain activation patterns.
Clinical Presentation of Oesophageal Complications
- Cardinal Symptoms: The primary oesophageal symptoms include reflux, heartburn, and dysphagia, though these are not specific to diabetic oesophageal dysfunction
- Gastroesophageal Reflux: Gastroesophageal reflux disease, including erosive oesophagitis, occurs more frequently in individuals with diabetes than in the general population.
- Potential Complications: While not firmly established, this may increase the risk of Barrett’s oesophagus and therefore oesophageal adenocarcinomas
Assessment and Management of Oesophageal Dysfunction
Symptomatic Evaluation
Assessment of gastro-oesophageal reflux disease often relies on the symptomatic presentation as the initial diagnostic approach.
- Endoscopic Examination: Oesophagogastroduodenoscopy is used to identify mucosal complications and rule out malignancies. When endoscopy is unrevealing, symptomatic therapies are often initiated.
- Advanced Testing: When symptomatic therapy is ineffective, pH/impedance monitoring may be diagnostically beneficial.
Manometric measures are relevant when suspecting motility disturbances, with high-resolution manometry providing the most comprehensive evaluation. - Treatment Approaches: Management follows standard regimes, including lifestyle changes and proton pump inhibitors. For motility disturbances associated with hypomotility, prokinetic drugs like metoclopramide, domperidone, and erythromycin may be used empirically.
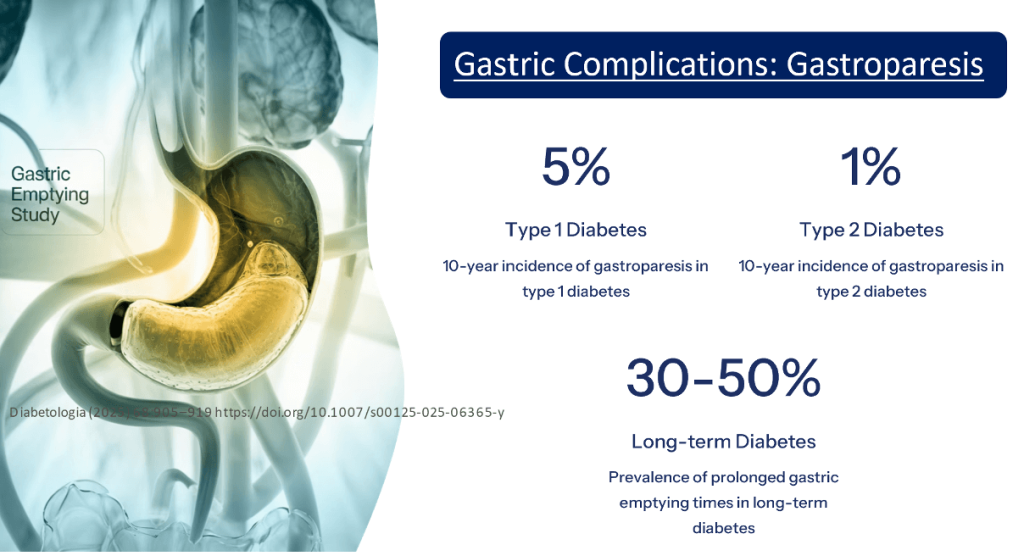
“Gastroparesis vs. Gastropathy”:
- Diabetic Gastroparesis: The best-described gastrointestinal complication, characterized by symptoms from the proximal gastrointestinal tract and delayed gastric emptying. Occurs more frequently in women and concomitantly with other diabetic complications.
- Diabetic Gastropathy: A more inclusive term, recognizing that symptoms can be attributed to diabetic gastrointestinal alterations regardless of whether the gastric emptying time is prolonged, normal, or rapid. The intraindividual gastric emptying time variation is considerable.
- Symptom Prevalence: Regardless of the underlying gastric emptying time, the prevalence of symptoms from the upper gastrointestinal tract in diabetes lies between 18% and 50%, with estimates varying widely across studies.
Pathophysiological Disturbances in the Diabetic Stomach
| Sensory dysfunction involves visceral hypersensitivity and central hyperexcitability, contributing to bloating and discomfort. |
| Impaired vagal afferent-efferent signalling is central to autonomic neuropathy affecting gastric function. |
| Postprandial motor dysfunction leads to impaired gastric accommodation and gastro-oesophageal reflux due to poor sphincter control. |
| Antral hypomotility and increased pyloric tone cause delayed gastric emptying and poor antral-duodenal coordination. |
| Intragastric motor dysfunction manifests as discoordinated contractions and disruption of fasting migrating motor complexes. |
| Loss of interstitial cells of Cajal (ICCs) affects pacemaker function, leading to dysmotility. |
| Diabetic gastropathy may involve either delayed or accelerated gastric emptying, not just gastroparesis. |
| Multiple contributors include autonomic neuropathy, ICC loss, structural remodelling, altered gut hormones, glycaemic variability, and certain glucose-lowering drugs. |
| Sympathetic and parasympathetic imbalance via vagus and celiac ganglia contributes to gastric dysregulation. |
| High-resolution manometry and gastric emptying studies are essential for comprehensive assessment |
Clinical Presentation of Gastropathy
- Cardinal symptoms of diabetic gastropathy include nausea, vomiting, bloating, fullness, and early satiety, often worsening after meals. Gastroparesis may also lead to gastro-oesophageal reflux.
- Symptom patterns are variable, with fluctuations in intensity and periodic exacerbations. In cases of accelerated gastric emptying, dumping syndrome may occur, presenting as early satiety, diarrhea, light-headedness, and postprandial hypoglycemia.
- Symptom-emptying discrepancy is common, as gastrointestinal symptoms do not always correlate well with gastric emptying times or accommodation measures. Delayed gastric emptying can occur without symptoms.
Complications of Abnormal Gastric Emptying
- Medication absorption is impaired in abnormal gastric emptying, potentially reducing the uptake of drugs and electrolytes and causing unpredictable therapeutic outcomes.
- Nutritional impact includes malnutrition and weight loss due to reduced dietary intake from symptoms, further complicating diabetes care.
- Glycemic control becomes challenging as inconsistent gastric emptying disrupts the synchrony between nutrient absorption and insulin action, leading to blood glucose fluctuations.
- Postprandial hypotension may result from impaired cardiovascular and gastrointestinal autonomic adaptation between fasting and fed states, presenting as dizziness or syncope after meals.
Assessment of Diabetic Gastropathy
| Symptomatic evaluation is crucial, and the Gastroparesis Cardinal Symptom Index (GCSI) questionnaire is a validated tool to assess upper gastrointestinal symptoms. |
| Endoscopic examination is often needed before evaluating gastric emptying to rule out other causes like obstruction or ulcers. |
| Gastric emptying studies such as scintigraphy, measuring radiolabeled meal retention at 2 and 4 hours, are considered the gold standard for evaluating gastric emptying time. |
| Alternative methods include gastric emptying breath tests, modified OGTTs for dumping syndrome, capsule-based technologies, and MRI, all of which can assess both gastric emptying and contractile function |
Basic Management of Diabetic Gastropathy
- Glycemic management involves continuous subcutaneous insulin infusion and glucose monitoring; hybrid closed-loop systems may be beneficial.
- Dietary modifications include a low-fiber, low-fat diet with small particle sizes consumed in small, frequent meals.
- Nutritional optimization focuses on correcting electrolyte and fluid imbalances, with nasogastric or jejunostomy feeding reserved for severe cases.
- Medication review requires reducing or discontinuing drugs that negatively affect gastrointestinal function.
Advanced Interventions for Refractory Gastroparesis
| Gastric neurostimulator implantation can relieve symptoms of gastroparesis and reduce the need for symptom-based pharmacotherapy in patients with drug-refractory disease. |
| Gastric peroral endoscopic myotomy (G-POEM) may improve symptoms in select patients by enhancing pyloric function and promoting gastric emptying. |
| Enteral feeding using a nasogastric tube or jejunostomy may be required in severe cases with substantial malnutrition or weight loss to support nutrition. |
| Evidence limitations exist, as current data on gastric neurostimulators and endoscopic interventions are limited, necessitating more robust research. |

Aetiologies of Diabetic Diarrhoea – Clinical Pearls
| Microbiota dysbiosis disrupts gut flora balance, contributing to chronic diarrhoea. |
| SIBO (Small Intestinal Bacterial Overgrowth) is common in diabetes and leads to bloating, malabsorption, and diarrhoea. |
| Coeliac disease may coexist with diabetes, especially Type 1, and must be ruled out. |
| Exocrine pancreatic insufficiency impairs digestion, causing steatorrhea and loose stools. |
| Postprandial cholecystoparesis leads to delayed bile release and fat malabsorption. |
| Bile acid malabsorption causes secretory diarrhoea and may mimic IBS-D. |
| Small intestinal and/or colonic dysmotility is driven by autonomic neuropathy. |
| Sphincter dysfunction with discoordinated defecation increases the risk of incontinence in diabetic patients. |
Small Intestinal Assessment
- GSRS questionnaire helps evaluate proximal and distal GI symptoms; use the Bristol Stool Form Scale for stool consistency.
- Scintigraphy can measure intestinal transit time but is limited by radiation exposure, time, and poor standardization.
- Advanced motility tests like manometry, 3D-transit system, and MRI offer transit and contractility insights but are not widely accessible.
- SIBO diagnosis is best confirmed by jejunal aspirate culture (gold standard), though breath tests (H₂/CH₄) are commonly used despite lower sensitivity.
Treatment of Small Intestinal Complications
- Glycemic control helps reduce small intestinal symptoms and slows autonomic neuropathy progression.
- Anti-diarrheal agents like loperamide and opium tincture target opioid receptors but may cause constipation.
- SIBO treatment commonly uses rifaximin; recurrent cases may need repeated antibiotics due to underlying dysmotility.
- Specific therapies for exocrine pancreatic insufficiency and bile acid malabsorption should be added when these are contributing factors.

Clinical Features of Colonic Dysfunction
- Symptom Fluctuations: Patients may experience periods of severe constipation alternating with normal bowel movements or diarrhea.
- Overflow Diarrhea: Liquid stool may pass around impacted fecal matter, mimicking primary diarrhea.
- Overlapping Symptoms: Constipation-induced bloating, nausea, fullness, and abdominal discomfort may resemble gastroparesis.
- Medication Effects: Glucose-lowering drugs or those used for managing diabetes complications may worsen gastrointestinal symptoms.
Assessment Steps for Colonic Dysfunction
| Symptom Assessment: Use GSRS (Gastrointestinal Symptom Rating Scale) and Bristol Stool Scale to evaluate bowel symptom severity and patterns. |
| Medication Review: Identify drugs that may be causing or worsening constipation. |
| Transit Time Measurement: Use radiopaque markers to assess for slow-transit constipation. |
| Anorectal Testing: Consider when a defecatory disorder is suspected. |
Treatment Approaches for Colonic Dysfunction
- Hydration: Adequate fluid intake helps soften stool consistency.
- Physical Activity: Regular exercise stimulates colonic motility.
- Dietary Fiber: A balanced fiber approach is needed, especially when gastroparesis coexists—dietitian input is crucial.
- Laxative Therapy: Osmotic and peristaltic agents can be used to relieve constipation symptoms.
Note: Lifestyle modifications are essential but must be tailored to avoid conflict with gastroparesis dietary restrictions.
Prevalence of Fecal Incontinence in Diabetes
- 2.6% – General diabetic population: Baseline prevalence of fecal incontinence.
- 9% – Diabetics with complications: Higher prevalence in those with comorbidities.
- 20% – Symptomatic diabetic cohorts: Seen in selected patient populations with anorectal symptoms.
- 0.8% – Non-diabetic controls: Serves as the baseline comparison rate.
Key Mechanisms in Diabetic Anorectal Dysfunction
- Sphincter Coordination
- Diabetic autonomic neuropathy can impair coordination between internal and external anal sphincters, disrupting normal defecation control.
- Rectal Sensitivity
- Rectal hyposensitivity occurs in both early and long-standing diabetes, often seen with sphincter dysfunction, contributing to fecal incontinence.
- Defecation Mechanics
- Dyscoordinated defecation may worsen diabetic constipation and reflects a complex interplay of anorectal dysfunction that may present as constipation or incontinence.
Key Clinical Features of Diabetic Anorectal Dysfunction
- Fecal Incontinence Patterns
- Common with diabetic diarrhea when anorectal dysfunction is present.
- Passive incontinence can occur due to overflow from constipation.
- Timing of Symptoms
- Incontinence often occurs during sleep.
- May present as urge or passive incontinence, affecting quality of life significantly.
- Constipation Presentation
- Anorectal dysfunction may also cause constipation, leading to patients alternating between constipation and incontinence over time.
Assessment and Treatment Steps
| Initial Assessment: Rule out other causes of diarrhea and incontinence through history, physical examination, and basic investigations. |
| Specialized Testing: Use anorectal manometry, defecography, endoanal imaging, and balloon distention tests as indicated. |
| First-Line Treatment: Implement lifestyle modifications, dietary changes, anti-diarrheal agents, and ensure complete rectal emptying. |
| Advanced Interventions: Consider pelvic floor biofeedback, transanal irrigation, or sacral nerve stimulation for refractory cases. |
Prevalence of Exocrine Pancreatic Insufficiency (EPI) in Diabetes
- 33% – Median prevalence in Type 1 Diabetes
- 29% – Median prevalence in Type 2 Diabetes
Clinical Features of Exocrine Pancreatic Insufficiency
| Digestive Symptoms: Reduced pancreatic enzymes → malabsorption of nutrients and fat-soluble vitamins despite normal diet. |
| Gastrointestinal Manifestations: Symptoms include hunger or anorexia, steatorrhea, flatulence, bloating, and abdominal discomfort with weight loss and malnutrition. |
| Systemic Effects: Calcium and vitamin D malabsorption → decreased bone mineral density and related health issues. |
| Glycemic Impact: Pancreatic insufficiency may contribute to glycemic instability, perpetuating poor metabolic control. |
Gallbladder Pathophysiology in Diabetes
Key Points:
- Multifactorial Etiology:
- Gallbladder dysmotility in diabetes is linked to autonomic neuropathy and disease duration.
- Hormonal Factors:
- Reduced CCK sensitivity impairs gallbladder contractions and postprandial emptying.
- Primary Impairments:
- Diabetic gallbladder shows hypomotility, wall changes, and increased volume → leads to stasis, delayed emptying (cholecystoparesis), and gallstone risk.
Clinical Features of Gallbladder Dysfunction in Diabetes
- Gallstone Symptoms
- Gallstones may be asymptomatic or cause pain, typically in the upper right abdomen. Symptoms can be intermittent or chronic.
- Bile Acid Malabsorption
- Diabetic cholecystoparesis with impaired bile acid release post-meals can cause bile acid malabsorption → may lead to diarrhea or steatorrhea.
- Metabolic Factors
- Diabetes increases gallstone risk via associated obesity and dyslipidemia, contributing to biliary disease.
Gallbladder Dysfunction – Key Assessment & Management Points
| Ultrasonography: First-line, non-invasive tool for detecting stones and wall abnormalities. |
| Advanced Imaging: Use EUS, CT, or MRCP when ultrasound is inconclusive. |
| Metabolic Management: Control glycemia and lower cholesterol to address underlying metabolic contributors. |
| Prokinetic Therapy: Erythromycin may enhance gallbladder motility in severe hypomotility, though not standard for gallstones. |
CME INDIA Clinical Pearls – Diabetic Gastroenteropathy
- Widespread System Involvement: Diabetic gastroenteropathy affects the entire GI tract and related exocrine organs, necessitating a pan-systemic evaluation.
- Multifaceted Dysfunction: It involves global hypomotility, dyscoordinated GI contractions, impaired sensory signaling, and abnormal secretion—creating a complex clinical entity.
- Diagnostic Overlap: Symptom overlap from various GI regions complicates accurate diagnosis and requires careful clinical correlation.
- Research Imperative: Significant gaps remain in understanding and treating diabetic gastroenteropathy—highlighting the need for more targeted studies and personalized therapies.
References:
- Kornum, D.S., Krogh, K., Keller, J. et al. Diabetic gastroenteropathy: a pan-alimentary complication. Diabetologia68, 905–919 (2025). https://doi.org/10.1007/s00125-025-06365-y

Discover CME INDIA

- Explore CME INDIA Repository
- Examine CME INDIA Case Study
- Read History Today in Medicine
- Register for Future CMEs

Very very informative
Thnx
Very nice