CME INDIA Presentation by Dr. D. C. Tirupati Rao, MD, Consultant Physician, Chaitanya Hospital, Karimnagar, Telangana.
Based on a presentation at APICON-2026, Patna.

Abstract
- Diabetic kidney disease (DKD) remains one of the most powerful amplifiers of cardiovascular morbidity and premature mortality in people with type 2 diabetes mellitus (T2D).
- Despite decades of renin–angiotensin system (RAS) blockade and the recent transformative impact of sodium–glucose cotransporter-2 inhibitors (SGLT2i), a substantial burden of residual renal and cardiovascular risk persists.
- This residual risk is increasingly recognised to be driven by non-hemodynamic mechanisms—particularly inflammation, oxidative stress, and fibrosis—pathways insufficiently addressed by conventional therapies.
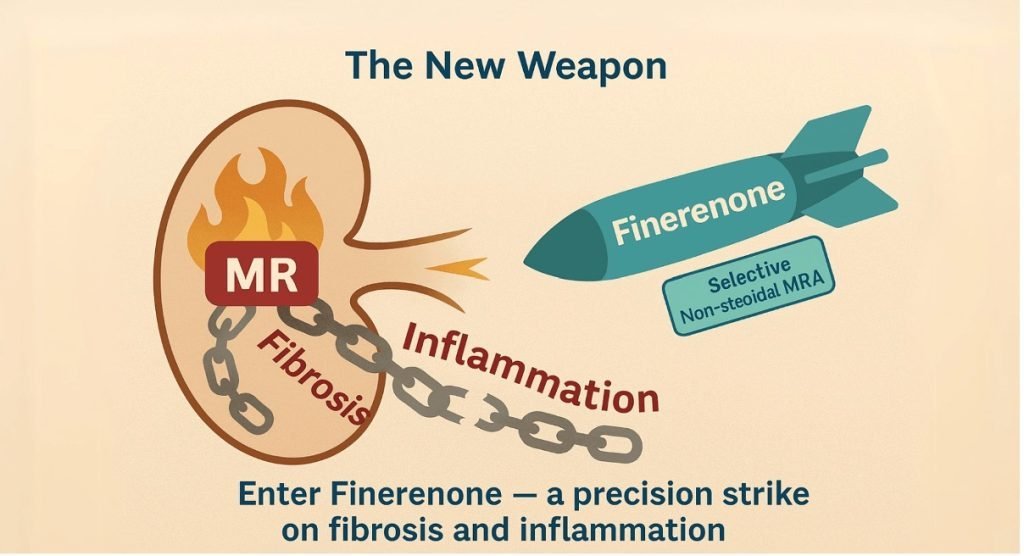
- Finerenone, a first-in-class non-steroidal mineralocorticoid receptor antagonist (nsMRA), has emerged as a targeted intervention against these mechanisms. Large cardiovascular-renal outcome trials, FIDELIO-DKD and FIGARO-DKD, together with the pooled FIDELITY analysis, have demonstrated consistent reductions in kidney failure progression, heart-failure hospitalisation, and cardiovascular events, with an acceptable safety profile.

- This review traces the evolving understanding of DKD pathobiology, examines the evidence supporting finerenone as a foundational therapy, and contextualises its role within contemporary guideline-directed, multi-pillar cardiorenal protection strategies.

1. The Unfinished Story of Diabetic Kidney Disease
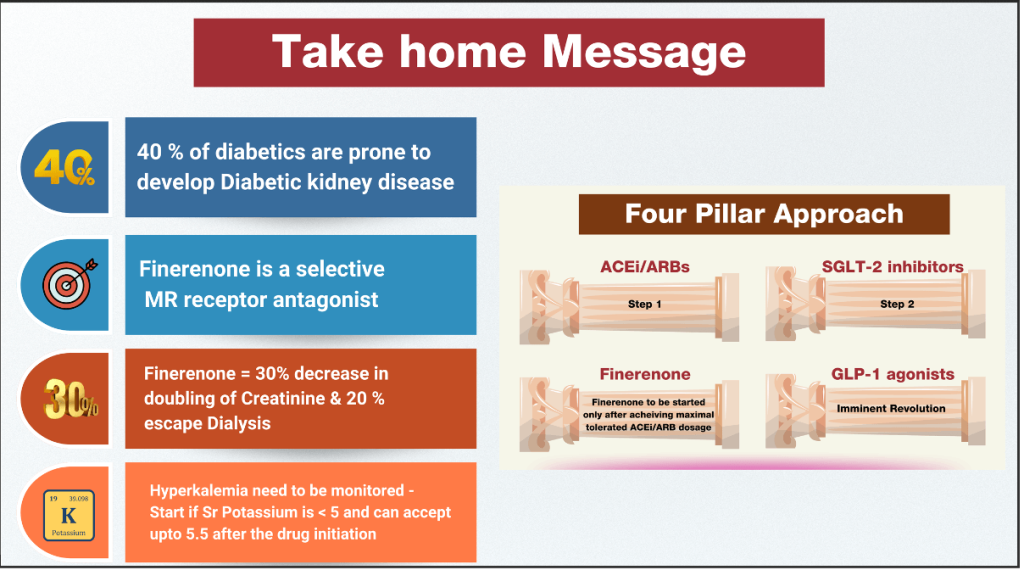
- Diabetic kidney disease affects approximately 20–40% of individuals with diabetes and remains the leading cause of chronic kidney disease (CKD) worldwide. Beyond the progressive loss of glomerular filtration rate (GFR), DKD fundamentally reshapes cardiovascular risk, conferring a 2- to 4-fold increase in cardiovascular mortality and substantially shortening life expectancy. Epidemiological analyses demonstrate that the combination of diabetes and CKD shortens life by up to 16 years—a loss exceeding that associated with either condition alone.
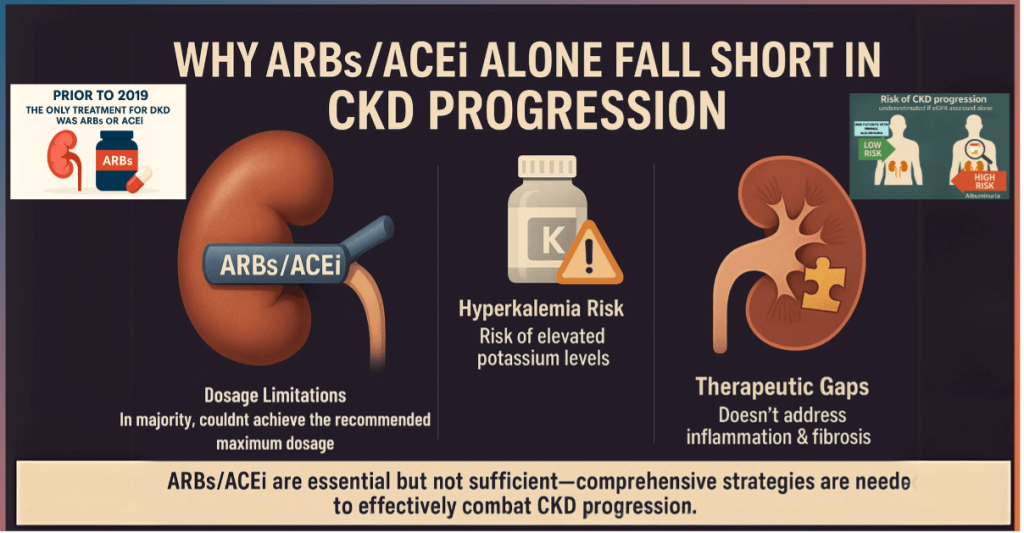
- For decades, DKD management revolved around glycaemic control and RAS inhibition using angiotensin-converting enzyme inhibitors (ACEi) or angiotensin receptor blockers (ARB). While these strategies reduce albuminuria and slow disease progression, they do not halt the inexorable decline in kidney function nor adequately mitigate cardiovascular risk. Even with optimal RAS blockade, many patients continue to progress toward end-stage kidney disease (ESKD), highlighting a fundamental therapeutic gap.

2. Albuminuria: More Than a Marker
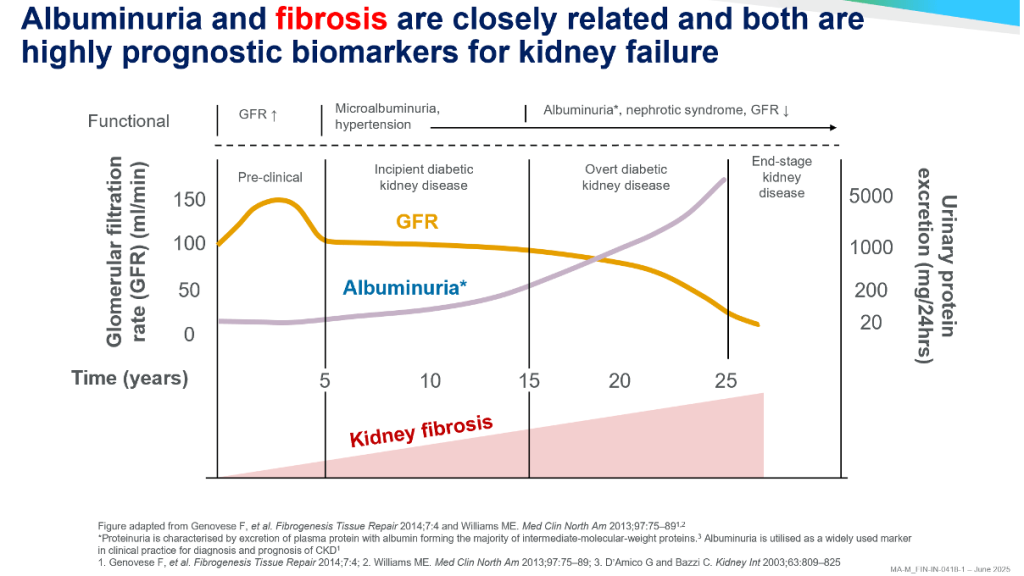
- Albuminuria has long been recognised as a diagnostic hallmark of DKD, but contemporary evidence positions it as a dynamic, prognostic, and therapeutic biomarker. Even mild elevations in urinary albumin-to-creatinine ratio (UACR ≥30 mg/g) are associated with graded increases in cardiovascular mortality and heart-failure risk. Importantly, albuminuria reflects ongoing glomerular inflammation, endothelial dysfunction, and tubulointerstitial injury rather than simple hemodynamic stress.

- Guidelines now emphasise that assessment of both UACR and estimated GFR (eGFR) is mandatory for accurate CKD staging and risk stratification. Reduction in albuminuria by ≥30% is recognised by regulatory agencies and professional societies as a meaningful surrogate endpoint for slowing DKD progression. Yet, traditional therapies achieve this target inconsistently, reinforcing the need for agents that directly modulate inflammatory and fibrotic pathways.

3. SGLT2 Inhibitors: A Transformative but Incomplete Revolution
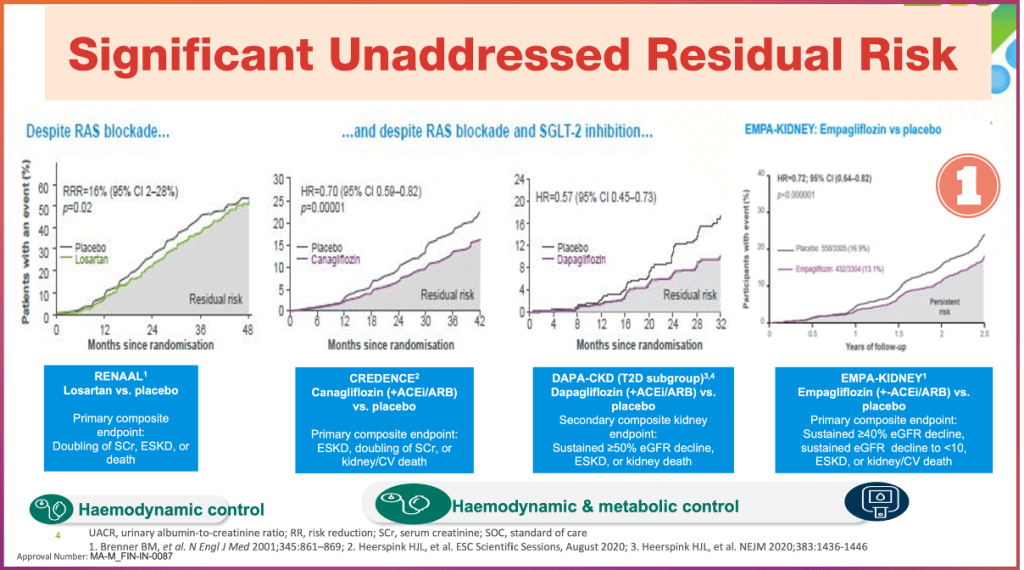
- The discovery that SGLT2 inhibitors confer robust renal and cardiovascular protection marked a paradigm shift in diabetes care. Trials such as CREDENCE, DAPA-CKD, and EMPA-KIDNEY demonstrated reductions of approximately 30% in kidney failure outcomes and significant decreases in heart-failure hospitalisation, independent of glucose lowering.
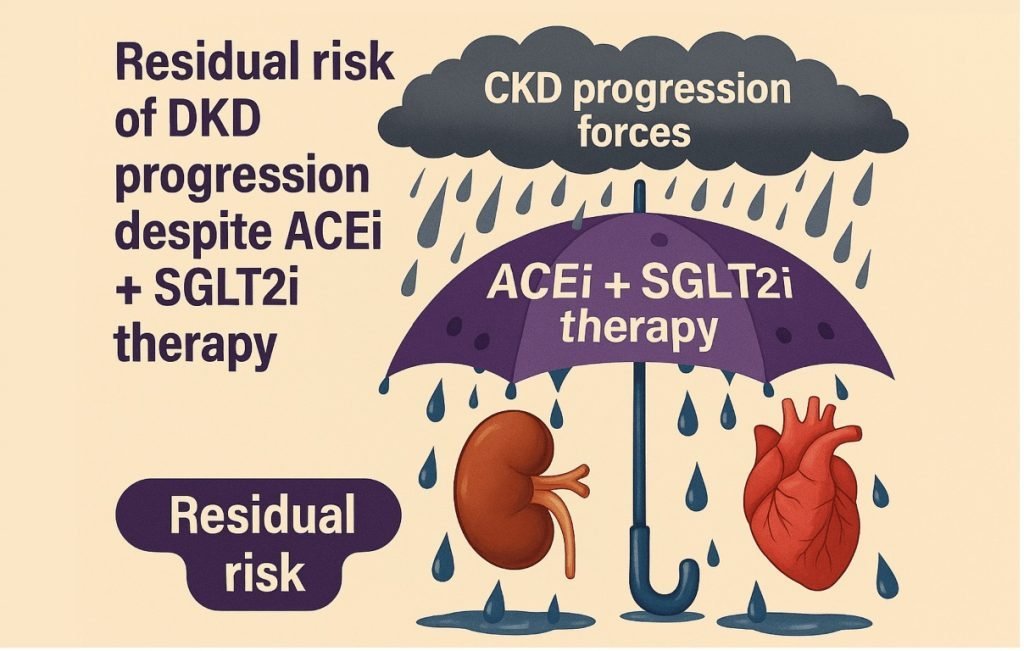
- However, despite these benefits, a substantial residual risk remains. Patients treated with ACEi/ARB plus SGLT2i continue to experience progressive albuminuria, declining GFR, and cardiovascular events. Mechanistically, SGLT2 inhibitors primarily address intraglomerular hypertension, metabolic stress, and neurohormonal modulation but exert limited direct effects on mineralocorticoid receptor–mediated inflammation and fibrosis.
- This recognition reframed DKD as a multi-pathway disease requiring layered, mechanism-specific therapies rather than reliance on a single “breakthrough” class.
4. Mineralocorticoid Receptor Overactivation: The Missing Link
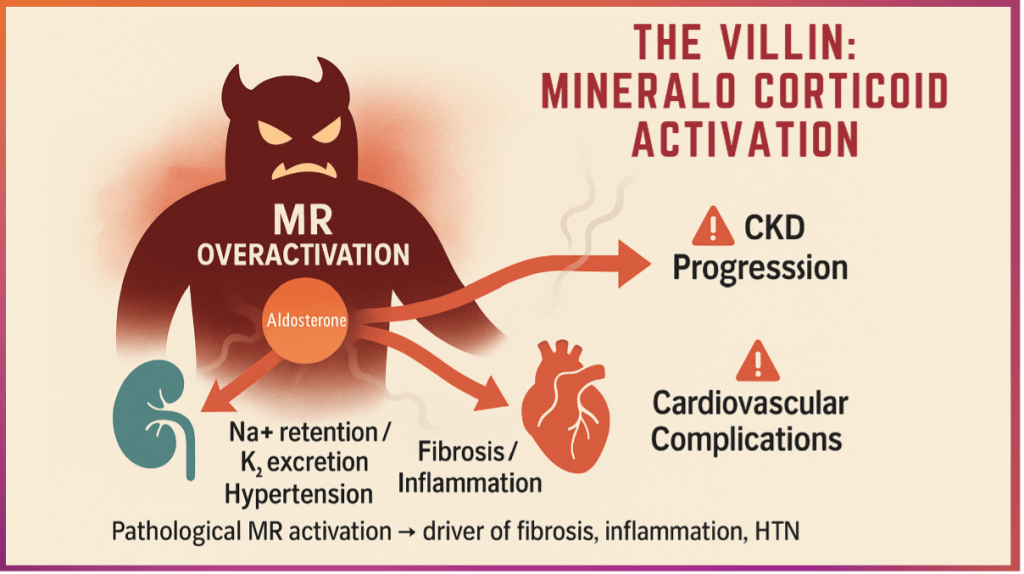
- Aldosterone and mineralocorticoid receptor (MR) activation play central roles in DKD progression. Beyond sodium retention and blood-pressure regulation, MR signalling promotes macrophage infiltration, oxidative stress, endothelial dysfunction, and profibrotic gene transcription in renal and cardiovascular tissues.
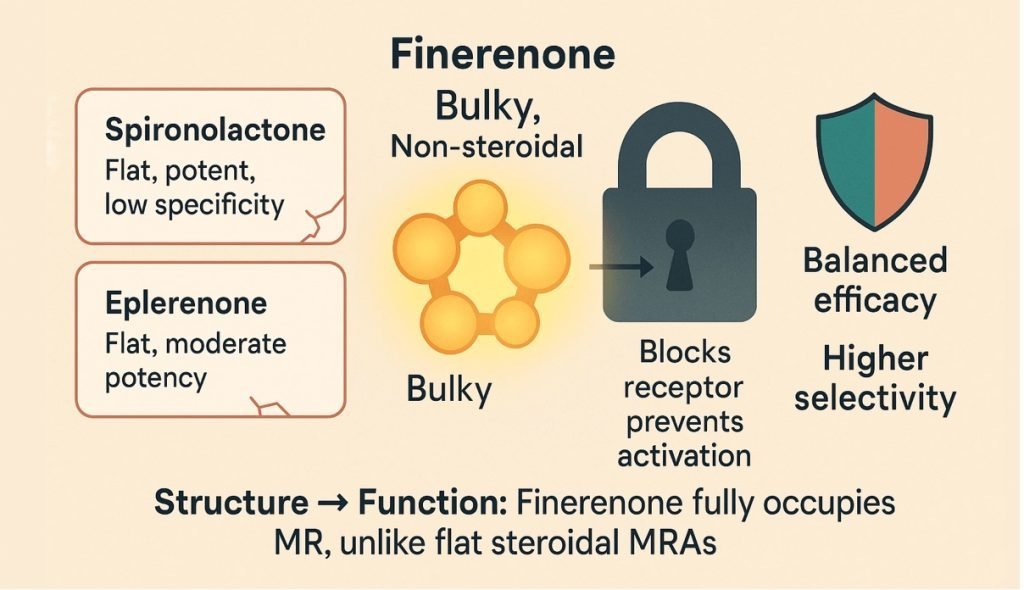
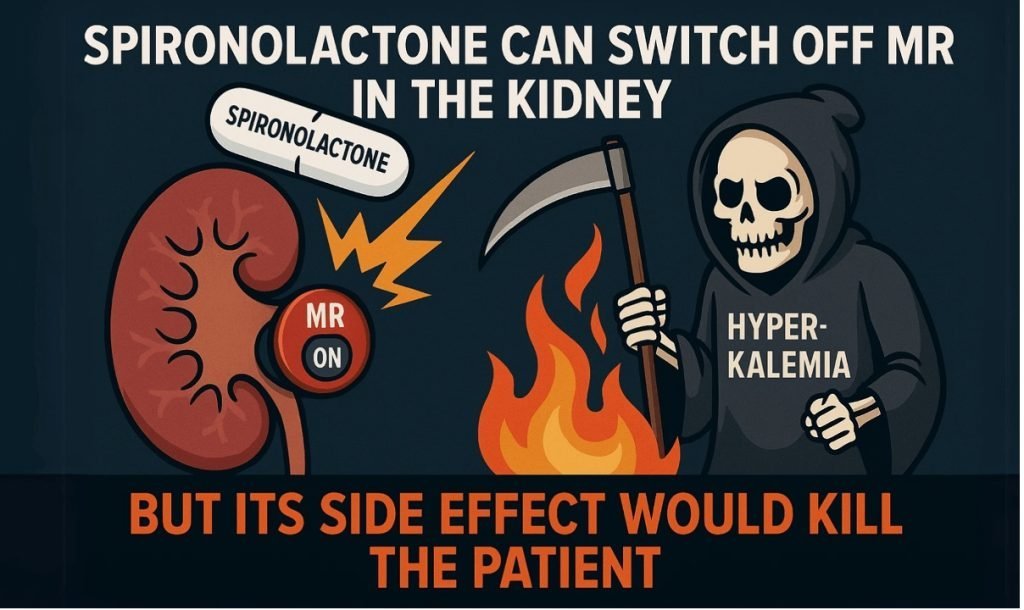
- Steroidal MR antagonists such as spironolactone and eplerenone reduce albuminuria but are limited by hyperkalaemia, gynecomastia, and renal adverse effects, particularly in patients with reduced eGFR. Consequently, their use in DKD has been restricted, leaving MR-mediated inflammation largely untreated in routine practice.
- Finerenone was developed to address this unmet need—a selective, non-steroidal MR antagonist with balanced cardiac and renal tissue distribution and a lower propensity for off-target endocrine effects.
5. Finerenone: Mechanistic Precision
- Finerenone binds the mineralocorticoid receptor with high affinity and induces a distinct conformational change compared with steroidal MRAs. This results in potent inhibition of inflammatory and fibrotic signalling pathways while exerting minimal effects on androgen and progesterone receptors.
- Preclinical models demonstrate that finerenone reduces renal macrophage infiltration, suppresses transforming growth factor-β–mediated fibrosis, and improves endothelial function. Importantly, these benefits occur with less potassium retention than seen with older MRAs, enabling safer use in CKD populations.
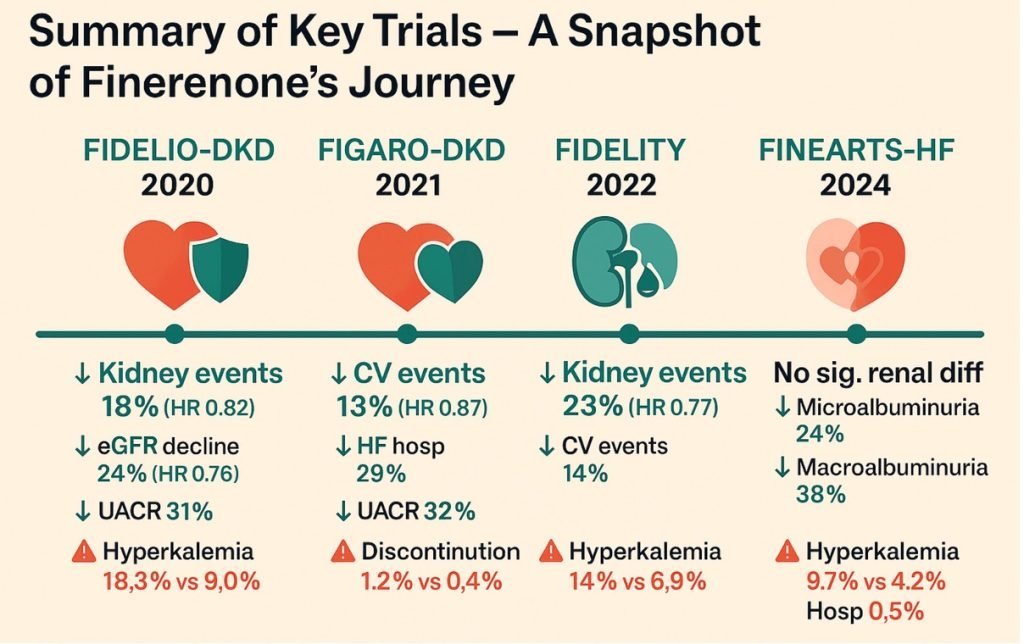
6. Landmark Clinical Trials: FIDELIO-DKD and FIGARO-DKD
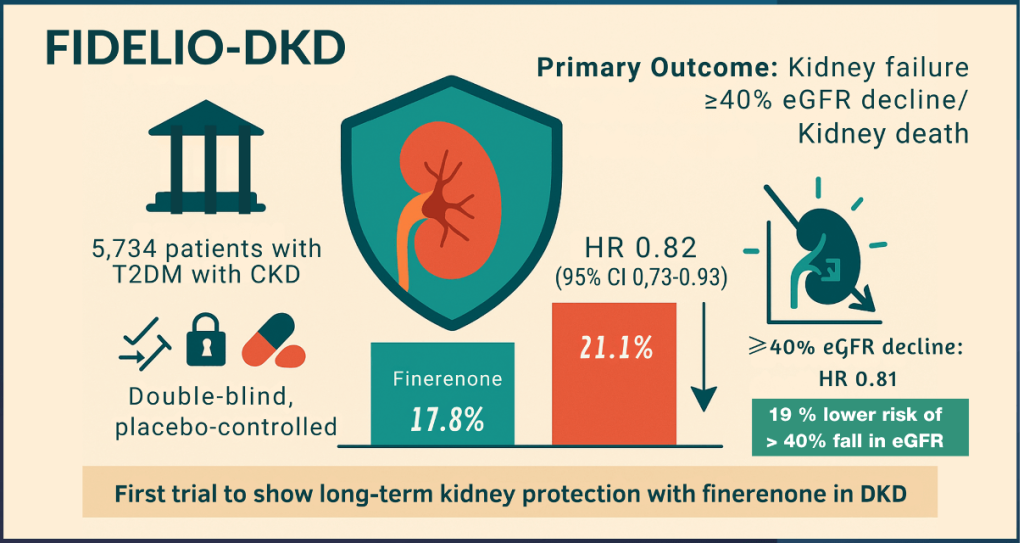
FIDELIO-DKD
- FIDELIO-DKD enrolled patients with T2D and advanced CKD with significant albuminuria, all receiving maximally tolerated ACEi or ARB therapy. Finerenone reduced the primary composite kidney outcome (kidney failure, sustained ≥40% decline in eGFR, or renal death) by 18% compared with placebo. Cardiovascular outcomes, particularly heart-failure hospitalisation, were also significantly reduced.
FIGARO-DKD
- FIGARO-DKD focused on patients with earlier-stage CKD and lower albuminuria. While renal outcomes were less frequent, finerenone significantly reduced the primary cardiovascular composite endpoint, driven largely by a reduction in heart-failure hospitalisation.
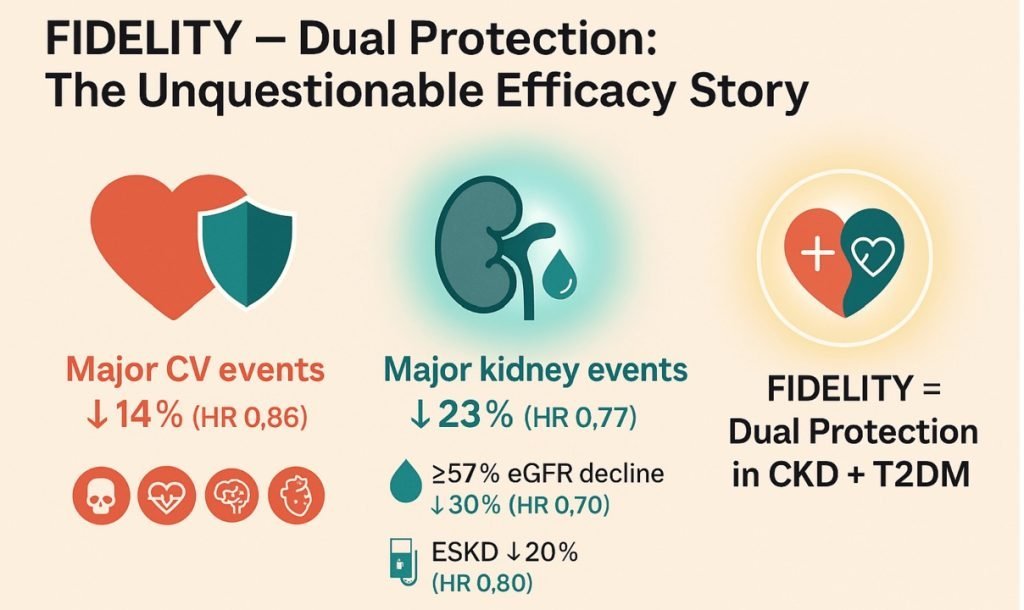
FIDELITY Pooled Analysis
- The prespecified pooled analysis of over 13,000 patients confirmed consistent cardiorenal benefit across CKD stages, with a ~23% reduction in kidney outcomes and ~14% reduction in cardiovascular events. Notably, benefits were observed regardless of baseline SGLT2 inhibitor use, supporting additive—not competitive—effects.
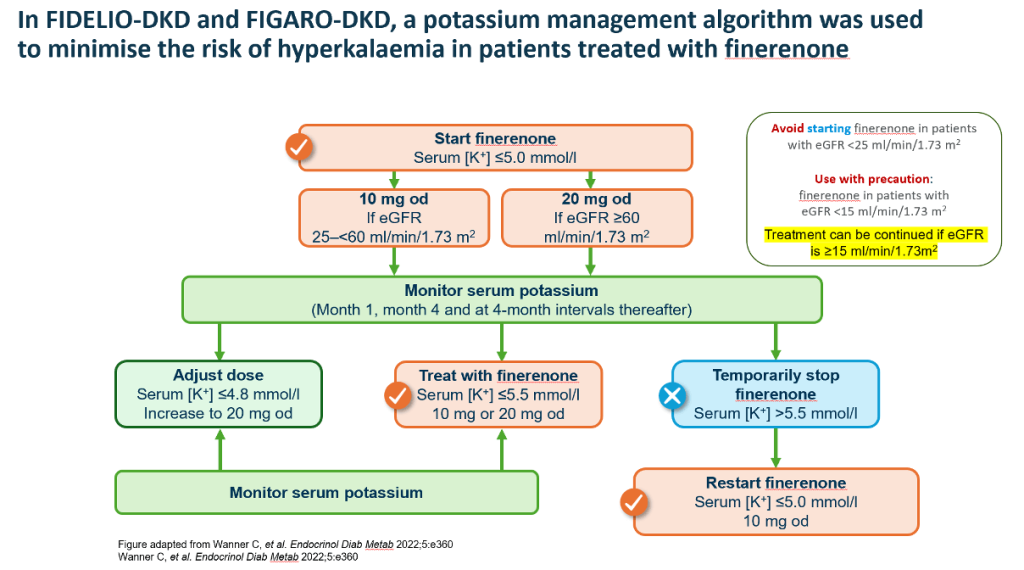
7. Safety and Potassium Management
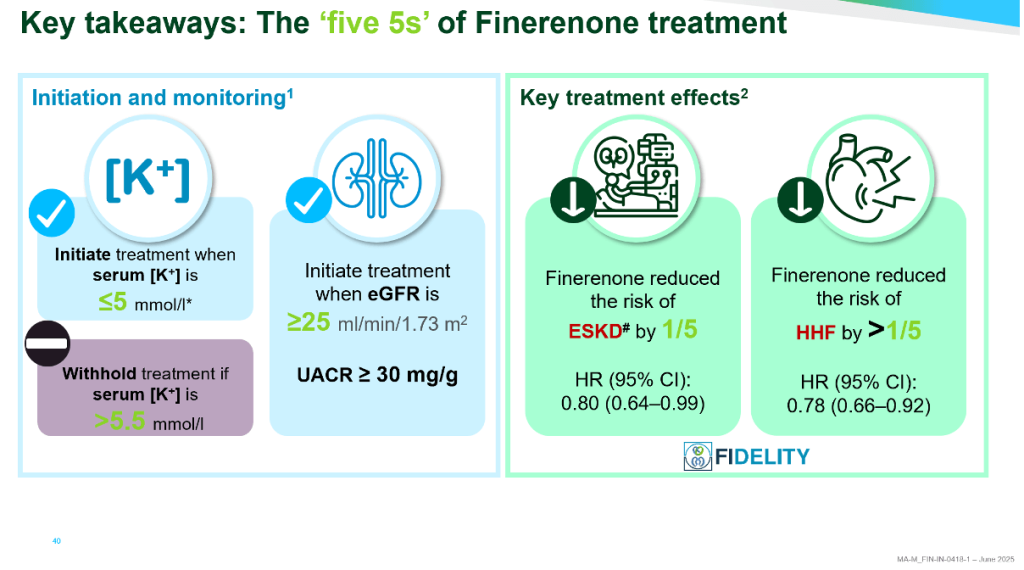
- Hyperkalaemia remains the principal safety consideration with finerenone, but trial-based potassium monitoring algorithms proved effective. Treatment discontinuation due to hyperkalaemia was infrequent, and serious adverse events were rare.
- Current guidance recommends initiating finerenone when serum potassium is ≤5.0 mmol/L and eGFR ≥25 mL/min/1.73 m², with structured potassium monitoring. Importantly, finerenone may be continued even if eGFR declines below initiation thresholds, reflecting its role in slowing disease progression rather than merely treating early disease.

8. Guideline Integration: From Evidence to Practice
- The 2022–2025 ADA–KDIGO consensus statements now recommend a three-pillar approach for DKD management in T2D:
- RAS inhibition (ACEi or ARB)
- SGLT2 inhibitor therapy
- Finerenone for patients with persistent albuminuria despite standard care
- This represents a decisive shift from glucose-centric management toward mechanism-based, organ-protective therapy. Finerenone is positioned not as a rescue drug but as a foundational component of comprehensive risk reduction.
9. Clinical Implications and Real-World Relevance
- In real-world practice, finerenone fills a critical therapeutic gap—particularly in patients with persistent albuminuria, declining eGFR, or high cardiovascular risk despite optimal ACEi/ARB and SGLT2 inhibitor therapy. Its glucose-independent benefits make it applicable across a wide glycaemic spectrum, including patients with near-target HbA1c.
- For clinicians, the challenge now lies not in evidence generation but in implementation: routine albuminuria screening, early identification of residual risk, and structured potassium monitoring are essential to unlock the full benefits of finerenone.
10. Conclusion

- Finerenone represents the culmination of a deeper understanding of DKD as an inflammatory-fibrotic disease rather than a purely hemodynamic complication of diabetes.
- By directly targeting mineralocorticoid receptor–mediated injury, finerenone complements existing therapies and meaningfully reduces both renal and cardiovascular events.
- As the therapeutic landscape evolves toward precision, pathway-driven care, finerenone stands as a cornerstone of modern cardiorenal protection in diabetes.
Key References:
- Bakris GL et al. Effect of Finerenone on Chronic Kidney Disease Outcomes in Type 2 Diabetes. N Engl J Med. 2020;383:2219-2229.
- Pitt B et al. Cardiovascular Events with Finerenone in Kidney Disease and Type 2 Diabetes. N Engl J Med. 2021;385:2252-2263.
- Filippatos G et al. Finerenone and Cardiovascular Outcomes in Patients With CKD and T2D (FIDELITY). Eur Heart J. 2022;43:474-484.
- KDIGO Diabetes Work Group. KDIGO 2022 Clinical Practice Guideline for Diabetes Management in CKD. Kidney Int. 2022;102:S1-S127.
- American Diabetes Association. Standards of Care in Diabetes—2025. Diabetes Care.2025;48(Suppl 1).
- Wanner C et al. Practical Potassium Management With Finerenone. Endocrinol Diabetes Metab. 2022;5:e00360.
- Agarwal R, Bakris GL. Nonsteroidal MRAs in Diabetic Kidney Disease. Nat Rev Nephrol. 2023;19:59-72.

Discover CME INDIA

- Explore CME INDIA Repository
- Examine CME INDIA Case Study
- Read History Today in Medicine
- Register for Future CMEs