CME INDIA Presentation by Dr. Parimal Swamy, MD, Diploma in Preventive Health Care (Apollo Hospital, Hyderabad), Consultant Physician JH & RC, Professor (Medicine) HIDS, Director Apollo Asthma & Diabetes Care Centre, Ex Member Executive Council M.P. Medical Science University.
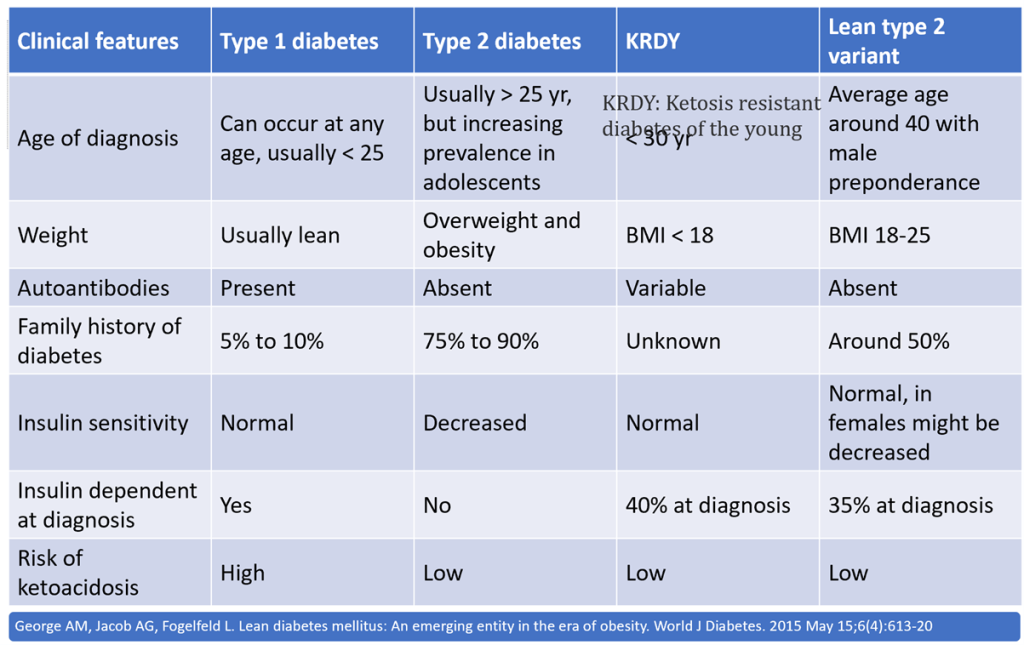
Spectrum of Lean Diabetics
- Low/normal body weight with abnormal (ectopic) fat deposition leading to early beta cell failure (pancreatic fat) at a lower insulin resistance (hepatic fat). (Insulin resistance is not high enough to cause generalized obesity) + Low muscle mass (sarcopenic obesity).
- LADA (Latent Autoimmune Diabetes of Adult)
- MODY (Maturity Onset Diabetes of the Young).
- Malnutrition (in utero/neonatal) related pancreatic dysfunction.
- Alcohol induced pancreatic dysfunction.
Early beta cell failure (pancreatic fat) at a lower insulin resistance (hepatic fat).
- To have the same diabetes risk as white participants with a BMI > 30 kg/m2, the equivalent BMI in South Asians was only 22 kg/m2.
- The underweight resulted a protective factor against T2D only in non-Asian people.
- The key feature of non-obese T2D seems to be a defect in the insulin secretion capacity as opposed to peripheral insulin resistance described in classical diabetes.
Here is the extracted and rewritten content from the image titled “Figure 1. Pathophysiological characteristics of lean Diabetes”:
Physiopathology of Non-Obese Type 2 Diabetes
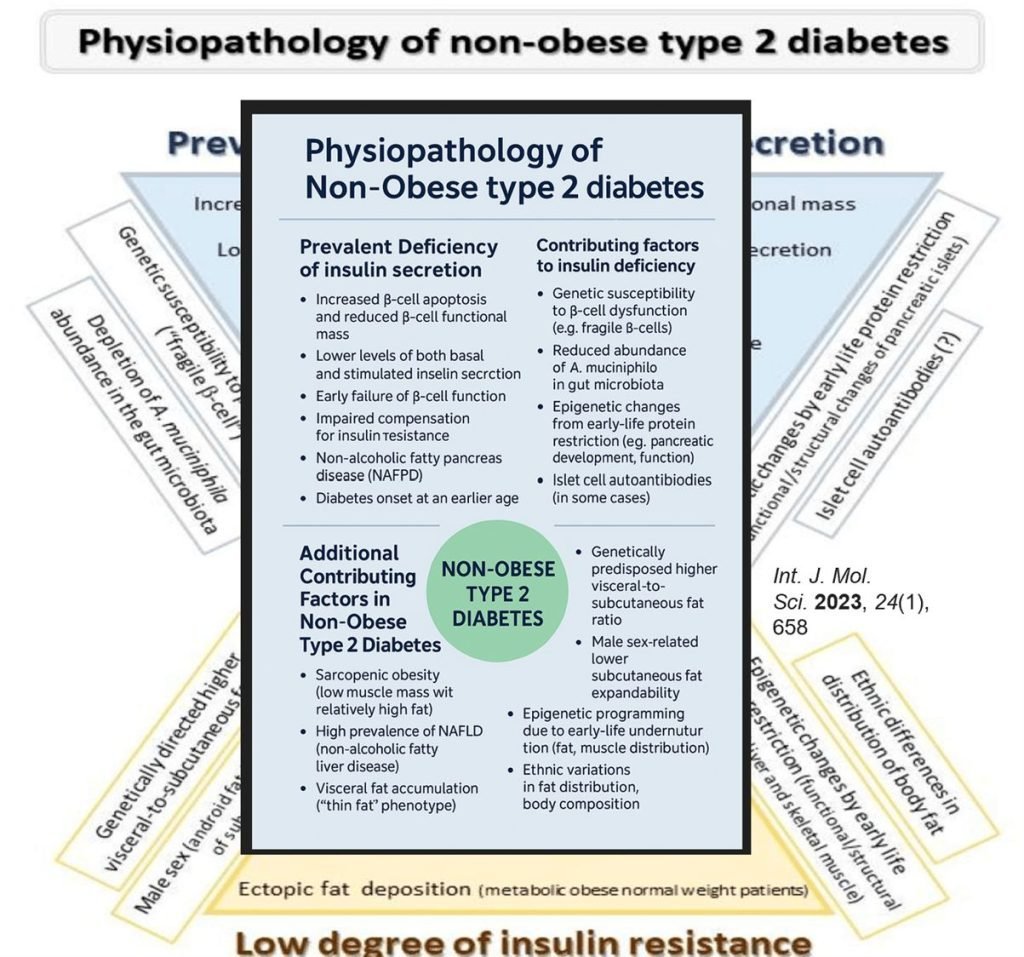
Central Feature: Prevalent Deficiency of Insulin Secretion
- Increased β-cell apoptosis and reduced β-cell functional mass.
- Lower basal and stimulated insulin secretion levels.
- Early β-cell failure.
- Impaired compensation for insulin resistance.
- Presence of non-alcoholic fatty pancreas disease (NAFPD).
- Onset of diabetes at a younger age.
Contributing Factors to Insulin Deficiency:
- Genetic susceptibility to β-cell dysfunction (e.g., fragile β-cells).
- Reduced abundance of A. muciniphila in the gut microbiota.
- Epigenetic changes from early-life protein restriction, affecting pancreatic development and function.
- Presence of islet cell autoantibodies (in some cases).
Additional Contributing Factors in Non-Obese Type 2 Diabetes
- Sarcopenic Obesity (low muscle mass with relatively high fat).
- High prevalence of NAFLD (non-alcoholic fatty liver disease).
- Visceral fat accumulation despite low overall adiposity (“thin fat” phenotype).
- Ectopic fat deposition, especially in the liver and muscle.
- Genetically predisposed higher visceral-to-subcutaneous fat ratio.
- Male sex-related lower subcutaneous fat expandability.
- Epigenetic programming due to early-life undernutrition, altering fat and muscle distribution.
- Ethnic variations in fat distribution and body composition.
Title: Developmental Origins of Lean Type 2 Diabetes
Key Concept: Maternal Low Protein Diet → Long-Term Metabolic Risk
| Pathway |
| 1. Maternal Low Protein Diet: |
| Leads to decreased amino acids in maternal blood. |
| 2. Nutritional Imbalance: |
| The amino acid supply fails to meet fetal requirements. |
| Results in intrauterine growth restriction (IUGR). |
| 3. Fetal Adaptation: |
| Metabolic changes occur in: |
| Muscle |
| Liver |
| Pancreas |
| Gonads |
| Brain |
| These are driven by: |
| Epigenetic modifications |
| Adaptation to protein-deficient environment |
| 4. Growth Trajectory: |
| In utero: restricted growth |
| Post-natal: |
| Catch-up growth |
| Predisposition to Lean Type 2 Diabetes (T2D |
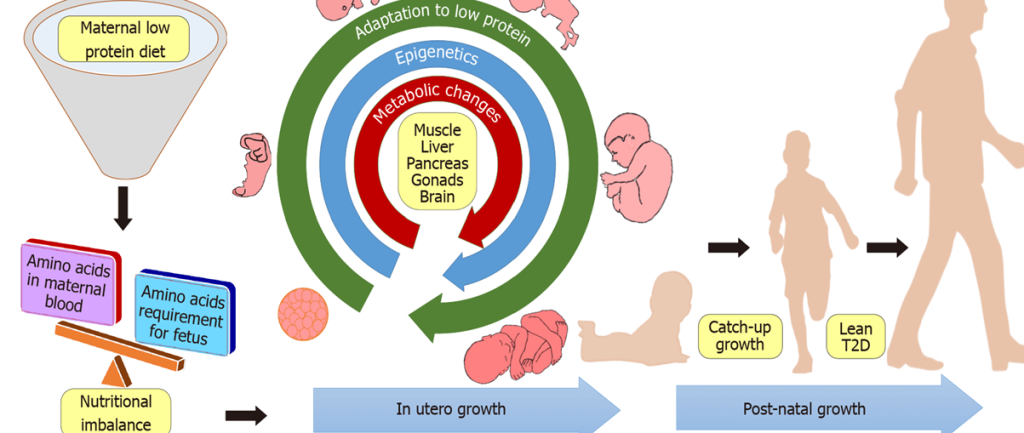
Courtesy: Vipin VA, Blesson CS, Yallampalli C. Maternal low protein diet and fetal programming of lean type 2 diabetes. World J Diabetes 2022; 13(3): 185-202
Lean Diabetes (patients with low-normal body mass index)
| History of childhood malnutrition, |
| Poor socioeconomic status |
| Early age of onset |
| Absence of ketosis |
| Males |
| Higher prevalence of smoking, alcoholism and pancreatitis |
| Higher prevalence of insulin use indicating rapid beta cell failure |
| Might have increased total, cardiovascular and non-cardiovascular mortality when compared to obese diabetic patients (western data) |
Pathophysiology: Malnutrition(!) (Alcohol) induced Pancreatic dysfunction
| Epigenetic changes due to maternal low protein diet |
| The key defect responsible for hyperglycemia in the lean diabetics is impaired pancreatic insulin secretion which is partly due to a reduced beta cell mass as demonstrated via autopsies. |
| The more severe beta cell dysfunction in these patients may be functional rather than structural as beta cell mass was noted to be equally reduced in both lean and obese patients |
| Chronic alcohol consumption induced pancreatic beta cell dysfunction and apoptosis |
| Genetic modulators might also predispose to reduced beta cell function in the lean body weight group |
Ketosis Resistant Diabetes of the Young (KRDY)
| (1) blood glucose > 200; |
| (2) onset < 30 years of age; |
| (3) BMI < 18 kg/m2; |
| (4) absence of ketosis on insulin withdrawal; |
| (5) poor socio-economic status or history of childhood malnutrition; |
| (6) insulin requirement > 60 units/d or 1.5 units/kg. |
Looking back: What was Indian Scenario :10000 type 2 diabetics
| 3.5% patients were lean with a BMI < 18.5 & 63% patients having ideal body weight at diagnosis. |
| HbA1c, fasting and postprandial blood glucose levels were higher among those in the lean group |
| Micro-vascular complications of Diabetes such as retinopathy, nephropathy and neuropathy were more common among the lean male patients |
| In whom (small number) C-peptide levels were measured, they were found to be significantly higher compared to the type 1 diabetics |
| Results of islet cell antibodies and antibodies against Glutamic acid decarboxylase were not significantly different between the 3 groups (lean/normal weight/obeseNearly 48% of the lean patients responded to diet or oral hypoglycemic agents after a mean duration of 9.2 ± 8.1 years, which is a clear distinction from type 1 diabetics. Symptomatic ketoacidosis was absent in this group |
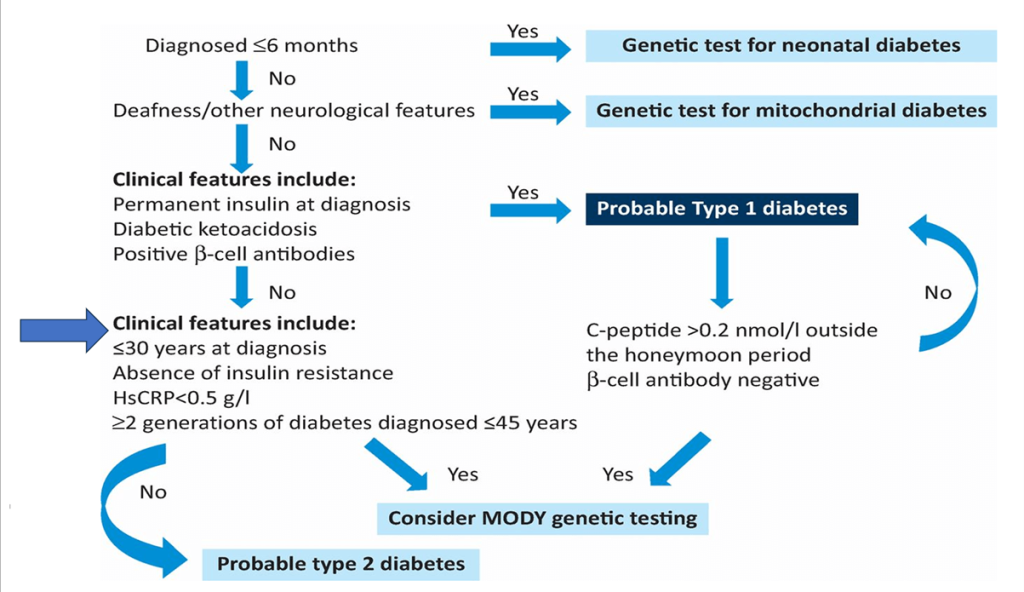
Evaluation
- History (MODY/Classical Type 2/’MRDM’)
- Autoantibodies
- C-Peptide
CME INDIA Learning Points
- Distinct Pathophysiology: Lean diabetes in Indians often involves early β-cell dysfunction with relatively less insulin resistance, differing from the classical type 2 diabetes phenotype seen in the West.
- Non-obese ≠ Non-diabetic: Even in the absence of obesity (BMI < 25 kg/m²), Indians may develop diabetes due to increased visceral fat, ectopic fat deposition (liver, pancreas), and genetic susceptibility.
- Ethnic and Genetic Factors: South Asians have a unique metabolic profile—reduced insulin secretion, low muscle mass, and increased hepatic fat—requiring ethnically tailored diagnostic and therapeutic approaches.
- Need for Early Screening: Traditional BMI cut-offs may miss high-risk individuals. Waist circumference, body composition, and metabolic markers should be prioritized for early detection.
- Dietary and Lifestyle Focus: Intervention should emphasize culturally adapted dietary modifications (e.g., low glycemic index foods, high fiber) and increased physical activity to improve insulin sensitivity and preserve β-cell function.
- Precision Medicine is Key: Lean diabetes may include cases of atypical diabetes (e.g., MODY, LADA). Genetic testing and antibody screening should be considered where appropriate to personalize treatment.
- Therapeutic Nuances: Over-reliance on insulin sensitizers like metformin may not be ideal in lean patients. Agents that preserve or enhance β-cell function (e.g., DPP-4 inhibitors, GLP-1 RAs) may be more beneficial.
- Undernutrition-Diabetes Link: A history of early life undernutrition or intrauterine growth restriction contributes to lean diabetes, reinforcing the need for a life-course approach to prevention.
- Public Health Implication: There is an urgent need to revise public health messaging and screening strategies in India to capture lean individuals at risk and avoid underdiagnosis.
- Further Research Needed: More large-scale Indian cohort studies are required to refine diagnostic criteria, understand subtypes, and optimize treatment algorithms for lean diabetes.
References:
- Salvatore T, Galiero R, Caturano A, Rinaldi L, Criscuolo L, Di Martino A, Albanese G, Vetrano E, Catalini C, Sardu C, et al. Current Knowledge on the Pathophysiology of Lean/Normal-Weight Type 2 Diabetes. International Journal of Molecular Sciences. 2023; 24(1):658. https://doi.org/10.3390/ijms24010658
- Vipin VA, Blesson CS, Yallampalli C. Maternal low protein diet and fetal programming of lean type 2 diabetes. World J Diabetes 2022; 13(3): 185-202 URL: https://www.wjgnet.com/1948-9358/full/v13/i3/185.htm DOI: https://dx.doi.org/10.4239/wjd.v13.i3.185
- George AM, Jacob AG, Fogelfeld L. Lean diabetes mellitus: An emerging entity in the era of obesity. World J Diabetes. 2015 May 15;6(4):613-20
- Ahuja MM. Diabetes-special problems in developing countries. Bull Deliv Health Care Diabetics Devel Countries. 1980;1:5–6 https://ouci.dntb.gov.ua/en/works/4YzQa26l/
- Mohan V, Vijayaprabha R, Rema M, Premalatha G, Poongothai S, Deepa R, Bhatia E, Mackay IR, Zimmet P. Clinical profile of lean NIDDM in South India. Diabetes Res Clin Pract. 1997;38:101–108.

Discover CME INDIA

- Explore CME INDIA Repository
- Examine CME INDIA Case Study
- Read History Today in Medicine
- Register for Future CMEs


A very apt description of Lean Type 2 DM covering all aspects ranging from pathophysiology to other contributory factors in causation of Lean Type 2 DM. Its important to keep in mind the possibility of LADA and MODY while entertaining the differential diagnosis.Inview of relatively preserved insulin sensitivity,the role of Metformin is limited and should not be prescribed,however DPP4 inhibitor and GLP1 Analog have a role to play in the treatment of Lean Type 2 DM patients.
Excellent presentation