CME INDIA Case Presentation by Dr. Kirti Soota, Diabetologist scope certified, Dr. Kirti Soota’s Diabetes and Obesity Clinic, New Delhi.
CME INDIA Case Study
How Presented?
- A 12-year-old girl with a history of childhood-onset obesity, neurodevelopmental delay, and severe insulin resistance.
- She had increased appetite and Grade 2 obesity. Homeostatic Model Assessment for Insulin Resistance (HOMA-IR) value showed severe insulin resistance apart from clinical pointers .She also suffered from OSA.

Let us see the result of whole Exome Sequencinge3zp96
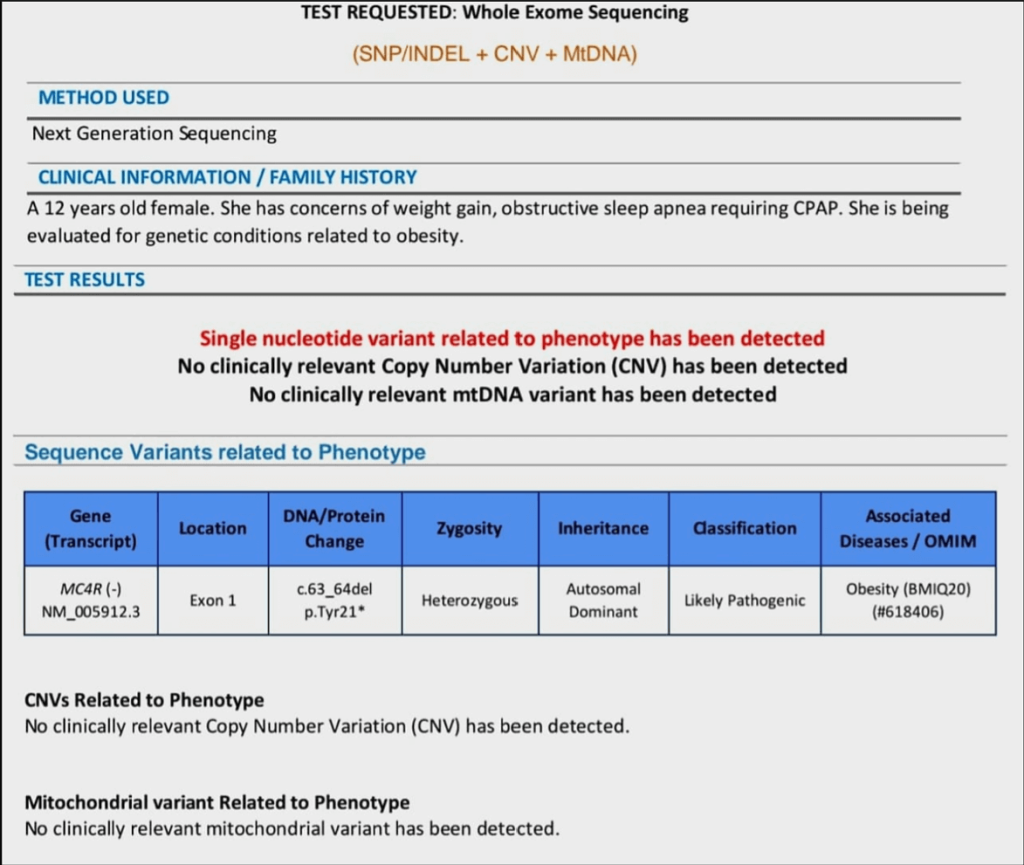
Key Findings from Report:
- Gene Involved: MC4R (Melanocortin 4 Receptor gene) Variant: c.63_64del (p.Tyr21*) → This is a nonsense mutation leading to a premature stop codon.
- Zygosity: Heterozygous Inheritance: Autosomal Dominant Classification: Likely Pathogenic Associated Disease: Obesity (BMIQ20), OMIM #618406 —
Why this is Significant?
- MC4R is the most common gene associated with monogenic obesity Mutations in MC4R are found in 2–5% of children with early-onset severe obesity. It plays a key role in hypothalamic appetite regulation and energy homeostasis.
- The variant is a likely pathogenic truncating mutation The p.Tyr21* change introduces a premature stop codon, leading to loss of protein function, a known mechanism in MC4R-linked obesity.
- Clinical context fits well Early-onset severe obesity Obstructive sleep apnea Insulin resistance likely No other CNVs or mtDNA variants detected, making this SNV the most likely genetic contributor.
When to Consider Genetic Testing in an Obese Patient
| 1. Early-Onset Obesity |
| Onset of obesity before 5 years of age |
| Rapid and excessive weight gain in infancy or early childhood |
| 2. Severe or Refractory Obesity |
| Body mass index (BMI) >97th or >120% of the 95th percentile for age and sex |
| Failure to respond to structured lifestyle or pharmacologic interventions |
| 3. Family History Suggestive of Mendelian Inheritance |
| First-degree relatives with early-onset or severe obesity |
| Positive family history of consanguinity or known genetic disorders |
| 4. Associated Neurodevelopmental Features |
| Global developmental delay or intellectual disability |
| Autism spectrum disorder, speech or motor delay |
| 5. Syndromic Features |
| Dysmorphic facial features |
| Polydactyly, genital anomalies, retinal dystrophy (e.g., Bardet-Biedl syndrome) |
| Hypotonia or feeding difficulties in infancy |
| 6. Endocrine or Metabolic Red Flags |
| Hyperphagia (uncontrollable appetite) |
| Hypogonadism, growth hormone deficiency, or adrenal insufficiency |
| Severe insulin resistance or early-onset type 2 diabetes |
| Acanthosis nigricans in very young children |
| 7. Suspected Monogenic Obesity Clues |
| Severe obesity with normal height and no syndromic features |
| Extreme hyperphagia with behavioral food-seeking |
| Known variants in obesity-related genes (MC4R, LEPR, POMC, PCSK1, etc.) |
Implications for Diagnosis & Management:
- Diagnosis This is monogenic obesity due to MC4R heterozygous loss-of-function variant. Family Testing Test parents and siblings due to autosomal dominant inheritance.
- Therapeutic Considerations Lifestyle + consider setmelanotide (MC4R agonist) in eligible cases (currently approved for biallelic POMC/LEPR/PCSK1; in trials for MC4R variants). Psychosocial Support Important to counsel that this is a biological, not behavioral condition. —
Knowing about Clinical Significance MC4R deficiency
The variant occurs at extremely low population frequencies (0.007% in South Asian and 0.006% in African populations), supporting its pathogenic potential.
Clinical Significance MC4R deficiency represents the most common monogenic cause of obesity*, accounting for 2-5% of severely obese children and approximately 1 in 500 individuals in the general population.
The clinical presentation in this case is entirely consistent with MC4R deficiency:
- Early-onset severe obesity with rapid weight gain.
- Obstructive sleep apnea requiring CPAP therapy.
- Metabolic complications including likely insulin resistance.
- Hyperphagia and increased appetite (typical but not explicitly documented in this report).
Pathophysiology and Inheritance
The MC4R protein is crucial for hypothalamic regulation of appetite and energy expenditure through the leptin-melanocortin pathway[. Loss-of-function variants result in:
- Impaired satiety signalling.
- Increased food-seeking behaviour.
- Accelerated linear growth in childhood.
- Hyperinsulinemia and metabolic dysfunction.
The autosomal dominant inheritance pattern means each offspring has a 50% risk of inheriting the variant, making family screening essential.
Management Recommendations
Immediate Clinical Actions:
- Family genetic counselling and cascade testing of parents and siblings.
- Multidisciplinary obesity management* team involvement.
- Metabolic screening including glucose tolerance testing and lipid profiles.
Treatment Considerations:
| Standard lifestyle interventions remain important but may have limited efficacy. |
| GLP-1 receptor agonists (liraglutide, semaglutide) have shown promise in MC4R deficiency, achieving 6-19% weight loss in clinical studies.GLP-1 receptor agonists have shown limited but interesting potential in certain forms of monogenic obesity, particularly where there is partial MC4R pathway activity preserved. In classic cases with complete loss-of-function mutations in POMC, LEPR, or MC4R, the response to GLP-1s is usually poor, as these agents act downstream of the disrupted melanocortin pathway. However, in atypical or milder genetic variants — or in patients with coexisting metabolic features — GLP-1s may offer modest appetite and weight control benefits. |
| Setmelanotide is currently approved for upstream pathway defects (POMC, LEPR, PCSK1) but clinical trials are ongoing for MC4R variants.That said, targeted therapies like Setmelanotide remain the gold standard where genetic confirmation of MC4R pathway defects exists. |
| Bariatric surgery may be considered in severe cases, though outcomes are variable in MC4R deficiency. |
Psychosocial Support:
- Patient and family education about the biological basis of obesity to reduce self-blame and stigma.
- Psychological support for managing the chronic nature of genetic obesity
- School accommodation discussions regarding physical limitations and peer interactions.
Prognosis and Monitoring
Patients with MC4R deficiency typically experience:
- Progressive weight gain throughout childhood and adolescence.
- Metabolic complications including type 2 diabetes, dyslipidemia, and cardiovascular risks.
- Reproductive considerations for future family planning given the genetic nature.
Long-term monitoring should include:
- Regular assessment of obesity-related comorbidities.
- Sleep study follow-up for OSA management.
- Endocrine evaluation for metabolic complications.
- Consideration of emerging therapies as they become available.
This genetic diagnosis fundamentally changes the clinical approach from treating “lifestyle-related” obesity to managing a chronic genetic condition requiring specialized, individualized care strategies and realistic expectations for treatment outcomes.
CME INDIA Learning Points
- Monogenic obesity represents a rare but clinically important subset of childhood obesity, typically characterized by early-onset, severe weight gain due to single-gene defects that disrupt central appetite and energy regulation. Among these, variants in the melanocortin-4 receptor (MC4R) gene are the most frequently implicated.
- In this case, a 12-year-old girl with longstanding obesity, severe insulin resistance, and obstructive sleep apnea requiring CPAP underwent whole exome sequencing to explore potential genetic underpinnings.
- The results revealed a heterozygous single nucleotide deletion in the MC4R gene—c.63_64del, resulting in a premature stop codon (p.Tyr21*). This truncating mutation leads to early termination of the receptor protein, rendering it non-functional. Classified as “likely pathogenic,” this variant provides a compelling explanation for the patient’s phenotype and is strongly consistent with MC4R-associated monogenic obesity (OMIM #618406).
- The presence of this MC4R mutation holds both diagnostic and therapeutic implications. It validates the clinical suspicion of monogenic obesity, supports family screening due to its autosomal dominant inheritance, and opens avenues for targeted intervention as new therapies—such as melanocortin receptor agonists—emerge in the therapeutic landscape.
CME INDIA Tail-Piece
1. What is Setmelanotide?
A selective melanocortin-4 receptor (MC4R) agonist designed to treat genetic forms of obesity caused by impaired MC4R signaling.
FDA-Approved Indications
- POMC deficiency obesity
- LEPR deficiency obesity
- Bardet-Biedl Syndrome (BBS)
- Approved for children ≥6 years and adults
Mechanism of Action
- MC4R is a key regulator of hunger and satiety in the hypothalamus.
- Setmelanotide restores MC4R pathway signaling.
Worth to know
| >10% weight loss sustained in POMC/LEPR deficiency |
| Significant reduction in hunger scores |
| Not yet approved by CDSCO (India’s drug regulator) |
| Currently not commercially available in India |
| Cost (Internationally) |
| Approximate US pricing: |
| $330,000–$450,000 per year (USD) (That’s ₹2.75–3.75 crore annually) |
| (Note: Dose is weight-based, costs vary by age/weight) |
2. Monogenic Obesity: Are We Missing the Diagnosis?
Insights from 521 Obese Patients via Exome Sequencing
(Künzel, R., Faust, H., Bundalian, L. et al. Detecting monogenic obesity: a systematic exome-wide workup of over 500 individuals. Int J Obes (2025). https://doi.org/10.1038/s41366-025-01819-0)
Key Findings:
| Diagnostic Yield |
| Monogenic obesity: 5.8% |
| Potentially obesogenic variant: 7.1% |
| Total with relevant findings: 12.9% |
| Higher Yield in Subgroups |
| Children with obesity: 6.3% yield |
| Syndromic obesity: 7.0% yield |
| Lower yield in severe obesity — unexpected! |
| Obesity Gene Panels Are Incomplete |
| 40% of detected monogenic variants were in genes not included in current gene panels |
| Whole-exome sequencing (WES) outperformed limited panels |
Clinical Pearls:
| Don’t restrict genetic testing to extreme obesity cases |
| Consider WES in syndromic/early-onset pediatric obesity |
| Expand gene panels to include syndromic obesity genes |
| Genetic insights can guide personalized care & family counseling |
Bottom Line:
Genetic testing in obesity reveals clinically actionable variants in ~13% of cases—WES > Panels. Broader genetic screening may redefine obesity care, especially in pediatric & syndromic patients.
References:
- Ten S, Maclaren N. Insulin resistance syndrome in children. J Clin Endocrinol Metab. 2004;89(6):2526–39. doi:10.1210/jc.2004-0072
- Shalitin S, Phillip M. Childhood obesity, growth and puberty. J Clin Res Pediatr Endocrinol. 2009;1(4):183–94. doi:10.4274/jcrpe.v1i4.183
- Semple RK, Savage DB, Cochran EK, Gorden P, O’Rahilly S. Genetic syndromes of severe insulin resistance. Endocr Rev. 2011;32(4):498–514. doi:10.1210/er.2010-0020
- Styne DM, Arslanian SA, Connor EL, Farooqi IS, Murad MH, Silverstein JH, et al. Pediatric obesity—assessment, treatment, and prevention: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2017;102(3):709–57. doi:10.1210/jc.2016-2573
- Dabelea D, Mayer-Davis EJ, Saydah S, Imperatore G, Linder B, Divers J, et al. Prevalence of type 2 diabetes and pre-diabetes among US adolescents and young adults, 2005–2016. JAMA. 2019;321(8):753–65. doi:10.1001/jama.2018.7123
- ClinVar Miner. Submissions for variant NM_005912.3(MC4R):c.63_64del (p.Tyr21_Arg22delinsTer) [Internet]. Available from: https://clinvarminer.genetics.utah.edu/submissions-by-variant/NM_005912.3(MC4R):c.63_64del%20(p.Tyr21_Arg22delinsTer)
- Antuna-Puente B, Poitou C, Schlegel N, et al. Liraglutide treatment in a morbidly obese adolescent with a MC4R mutation. J Clin Res Pediatr Endocrinol. 2023;15(2):234–8. Available from: https://pmc.ncbi.nlm.nih.gov/articles/PMC10234058/
- Turan S, Bereket A. Current treatments for patients with genetic obesity. J Clin Res Pediatr Endocrinol. 2023;15(3):245–52. doi:10.4274/jcrpe.galenos.2023.2023-3-2. Available from: https://www.jcrpe.org/articles/current-treatments-for-patients-with-genetic-obesity/doi/jcrpe.galenos.2023.2023-3-2
- Genomics Education Programme. Monogenic obesity – Knowledge Hub [Internet]. Available from: https://www.genomicseducation.hee.nhs.uk/genotes/knowledge-hub/monogenic-obesity/
- PreventionGenetics. Monogenic obesity via the MC4R gene test [Internet]. Available from: https://www.preventiongenetics.com/testInfo?val=Monogenic-Obesity-via-the-MC4R-Gene
- Elferink A, Huhne J, Hochberg Z, et al. Outcomes of bariatric surgery and pharmacotherapy in MC4R-deficient patients. Front Endocrinol. 2021;12:792354. doi:10.3389/fendo.2021.792354. Available from: https://www.frontiersin.org/articles/10.3389/fendo.2021.792354/full
- Clément K, Kühnen P, Hainer V, et al. MC4R variants modulate α-MSH and setmelanotide-induced weight loss. J Clin Endocrinol Metab. 2024;109(10):2452–61. doi:10.1210/clinem/dgad423. Available from: https://academic.oup.com/jcem/article/109/10/2452/7639413
- Collet TH, Dubern B, Mokrosinski J, et al. MC4R pathway dysfunction in obesity: patient implications. Front Endocrinol. 2020;11:3790. Available from: https://pmc.ncbi.nlm.nih.gov/articles/PMC7263790/
- Obesity Canada. Canadian Adult Obesity Clinical Practice Guidelines – Psychological Interventions. 2021 [Internet]. Available from: https://obesitycanada.ca/wp-content/uploads/2021/07/10-Psych-Interventions-2-v7-with-links-1.pdf
- NCBI ClinVar. NM_005912.3(MC4R):c.63_64del (p.Tyr21_Arg22delinsTer) [Internet]. Available from: https://www.ncbi.nlm.nih.gov/clinvar/RCV004017689/
- Martín J, et al. Identification of a rare pathogenic MC4R variant. Front Genet. 2020;11:608840. doi:10.3389/fgene.2020.608840. Available from: https://www.frontiersin.org/articles/10.3389/fgene.2020.608840/full
- Noor N, et al. Homozygous LEPR mutation as a cause of early-onset childhood obesity. Aga Khan Univ Scholarly Works [Internet]. Available from: https://scholars.aku.edu/en/publications/homozygous-lepr-mutation-as-a-cause-of-early-onset-childhood-obes
- Farooqi IS, O’Rahilly S. A novel MC4R gene mutation in a girl with severe childhood obesity. J Clin Endocrinol Metab. 2009;94(3):765–8. Available from: https://pubmed.ncbi.nlm.nih.gov/19214805/
- Frühbeck G, Salas-Salvadó J, Martínez JA. Obesity and the regulation of energy balance. Int J Obes Relat Metab Disord. 2003;27(9):1056–63. Available from: https://dadun.unav.edu/bitstream/10171/17942/1/IntJObes200327.pdf
- Athena Diagnostics. Early onset obesity (MC4R) DNA sequencing test [Internet]. Available from: https://www.athenadiagnostics.com/view-full-catalog/early-onset-obesity-mc4r-dna-sequencing-test1
- Künzel, R., Faust, H., Bundalian, L. et al. Detecting monogenic obesity: a systematic exome-wide workup of over 500 individuals. Int J Obes (2025). https://doi.org/10.1038/s41366-025-01819-0.
Discover CME INDIA:

- Explore CME INDIA Repository
- Examine CME INDIA Case Study
- Read History Today in Medicine
- Register for Future CMEs

