CME INDIA Presentation by Dr. Mayura Choudhari Kale, MBBS, D. Diabetology, DNB (Medicine) Consultant Diabetologist, Dr. Kale’s Diabetes and Psychiatry Clinic, Visiting Diabetologist Mahaveer Hospital, Chhatrapati Sambhajinagar (Aurangabad).

Abstract
The increasing global prevalence of type 2 diabetes and obesity has led to rising use of glucagon-like peptide-1 receptor agonists (GLP-1 RAs). As nearly half of pregnancies are unplanned, inadvertent exposure to these drugs is increasingly encountered. Preclinical animal studies raise concerns about reproductive toxicity, whereas emerging human data suggest no significant increase in major congenital malformations compared with insulin. This article reviews current evidence, evaluates pharmacological and mechanistic safety profiles, summarizes recent cohort studies, and discusses clinical management strategies for preconception counselling and inadvertent exposure.
Introduction
Type 2 diabetes mellitus (T2DM) and gestational diabetes mellitus (GDM) are growing challenges worldwide, including in India, where prevalence rates are steadily rising. Pre-gestational diabetes affects approximately 1–2% of pregnancies, while GDM prevalence ranges from 3.8–21%. GLP-1 RAs, widely prescribed for T2DM and obesity, are increasingly used by women of reproductive age. This creates a clinical dilemma during pregnancy and preconception care, as safety data have historically been limited due to the systematic exclusion of pregnant women from randomized controlled trials.
Mechanism of Action and Pharmacokinetics
GLP-1 RAs enhance glucose-dependent insulin secretion, suppress glucagon release, delay gastric emptying, and promote satiety. Their half-life ranges from 2–4 hours (short-acting agents such as exenatide, lixisenatide) to 5–7 days (long-acting agents such as liraglutide, dulaglutide, semaglutide). Placental transfer appears minimal, with large peptide structure and rapid clearance suggesting a low theoretical teratogenic risk.

The Mechanistic role of GLP-1 on the hypothalamic–pituitary–gonadal (HPG) axis and reproductive organs, integrating evidence from in vitro, preclinical, and clinical studies.
- Hypothalamus & Pituitary: GLP-1 enhances GnRH release via kisspeptin/GABA neurons, leading to increased LH and FSH secretion.
- Ovaries: GLP-1 reduces androgens, increases SHBG, improves PCOS morphology, and reduces ovarian inflammation.
- Endometrium: GLP-1 reduces fibrosis and inflammation, suggesting a protective role.
- Testes: GLP-1 reduces inflammation, indicating possible fertility benefits.
- Clinical implication: Beyond glycemic control and weight loss, GLP-1 may directly influence reproductive function and fertility.

Preclinical Evidence
Animal studies demonstrate reproductive toxicity, including skeletal and visceral abnormalities, dose-dependent growth restriction, and embryonic death. These findings have driven regulatory agencies to advise against GLP-1 RA use in pregnancy.
Human Evidence
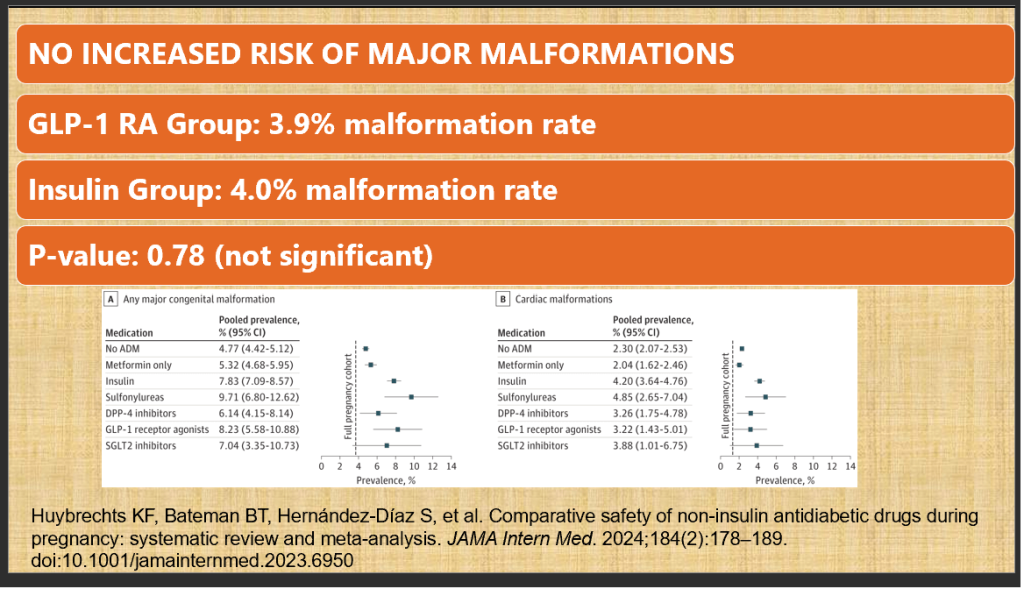
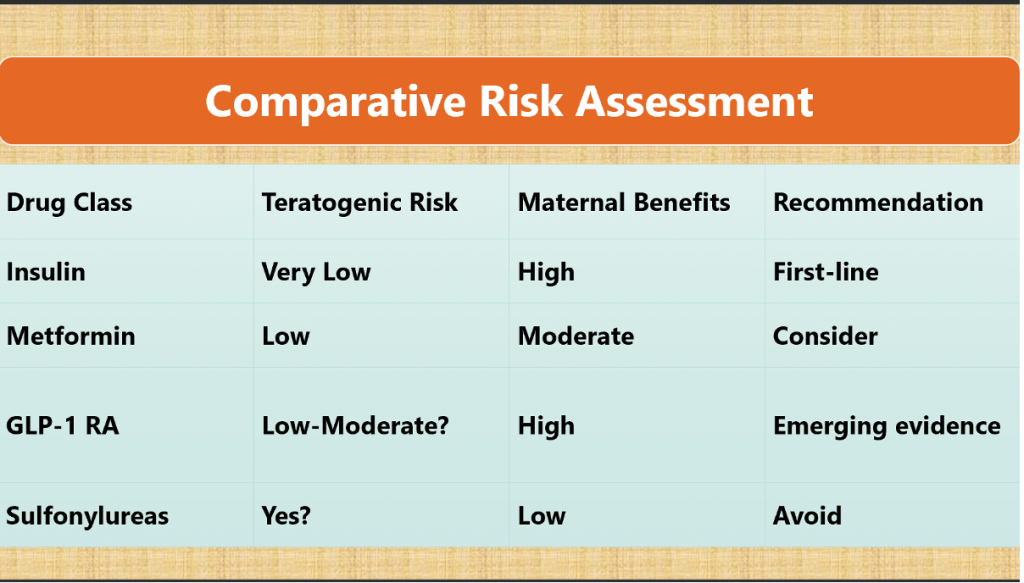
| Following figure reinforces that diabetes itself raises baseline malformation risk(insulin group ~7–8%, vs. general background 2–3%). |
| GLP-1 RAs do not appear riskier than insulin, but are not safer either — they cluster around the same range. |
| Metformin remains the most reassuring agent with no excess risk over baseline. |
| Sulfonylureas show the highest prevalence, suggesting avoidance in pregnancy. |
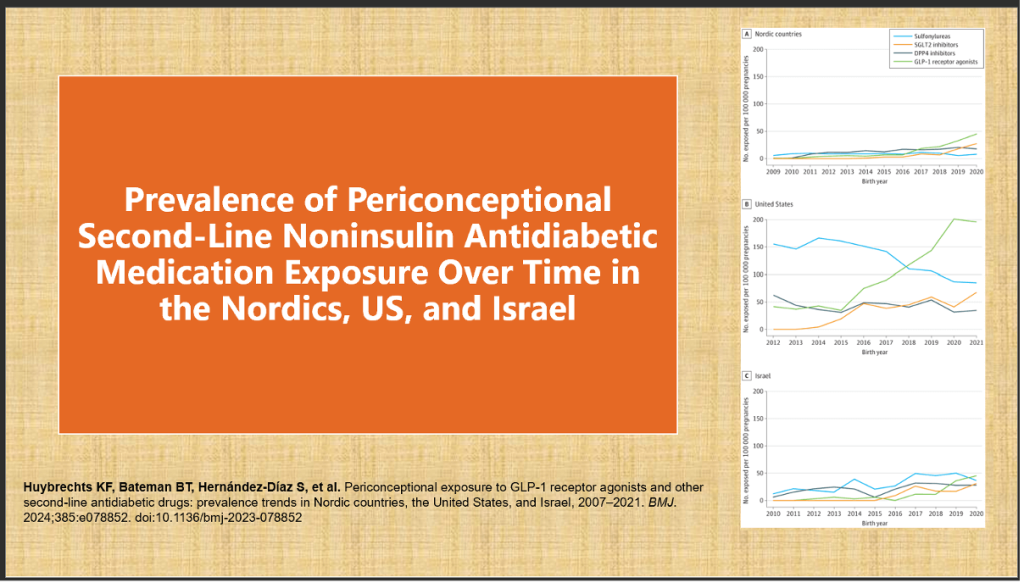
1. Multi-National Population-Based Cohort Study (Nordics + US, 2007–2020)
- Sample: 50,059 pregnancies (GLP-1 RA: 2,740; Insulin: 47,319)
- Primary outcome: Major congenital malformations
- Result: No significant increase in malformation risk (3.9% vs. 4.0%; p=0.78).
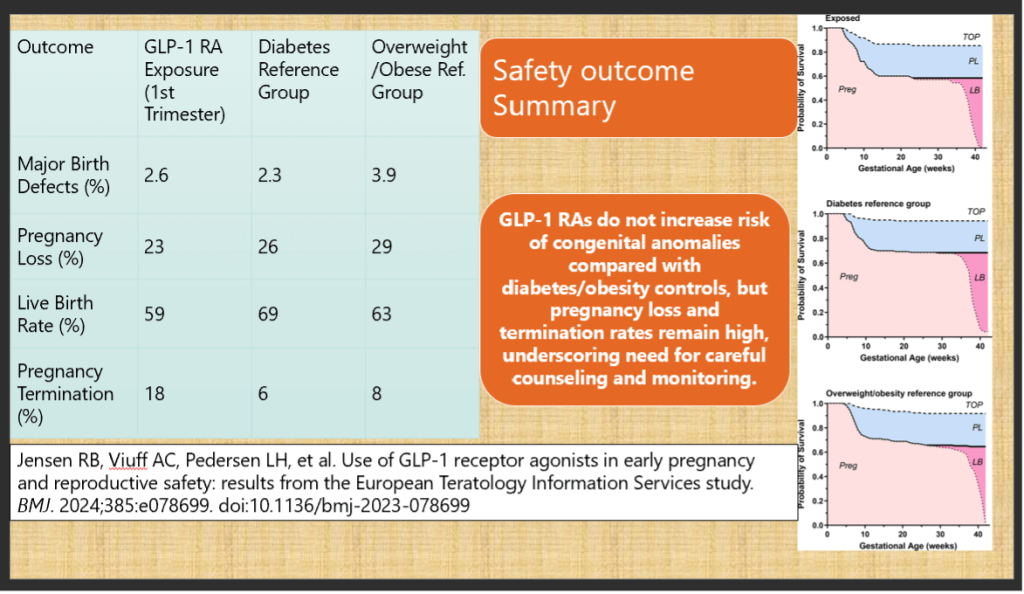
2. European Teratology Information Services Study (BMJ 2024)
- Design: Prospective observational cohort
- Sample: 168 GLP-1 RA-exposed pregnancies, compared with diabetic and obese reference groups
- Findings:
- Major birth defect rates: 2.6% (GLP-1 RA) vs. 2.3% (diabetes) vs. 3.9% (obesity)
- Pregnancy loss higher in all groups with metabolic risk (23–29%)
- Live birth rate: 59% (GLP-1 RA group)
- Conclusion: Reassuring data with no signal for increased congenital malformations.

Clinical Scenarios
Preconception Planning
- Transition to insulin 2–3 months prior to conception.
- Optimize HbA1c (<6.5%) before conception.
- Initiate folate supplementation.
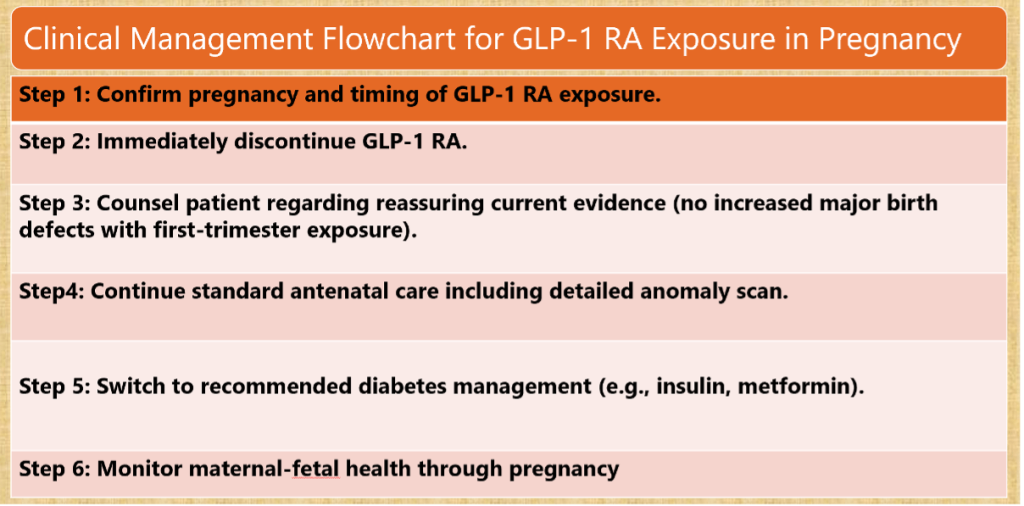
Inadvertent Exposure
- Immediate discontinuation of GLP-1 RA.
- Reassure based on current evidence (no excess malformation risk).
- Intensified monitoring with detailed ultrasonography.
- Switch to insulin or metformin.

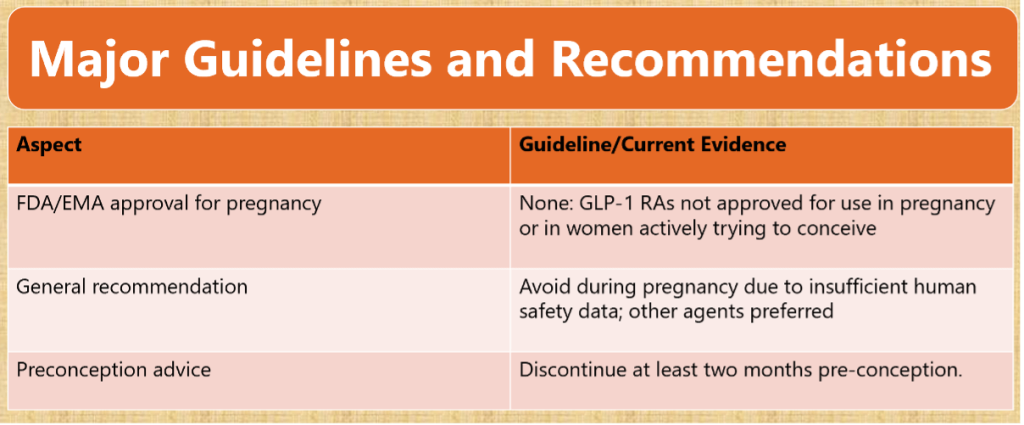
Guidelines and Recommendations
- Regulatory status: Not licensed for use in pregnancy (FDA, EMA).
- Professional societies: Recommend discontinuation at least 2 months prior to conception.
- Preferred therapy: Insulin remains the gold standard; metformin may be considered.
Scenario 1: Pre-conception Planning
28-year-old woman, BMI 26 kg/m2with T2DM on Metformin and liraglutide, planning pregnancy. Well controlled HbA1c 6%
Management Approach-
- Pre-conception counseling (3-6 months before conception).
- Risk-benefit discussion of medication options.
- Transition plan to insulin / discontinuing GLP-1 RA.
- Optimization of glycemic control before conception.
- Folate supplementation (5mg daily).

Scenario 2: Inadvertent Exposure
32-year-old woman with T2DM on semaglutide, presents at 8 weeks gestation
G2P0L0A1.First trimester pregnancy loss due to high blood glucose levels
Management- Stop Sema
- Reassurance based on current evidence
- Detailed risk counseling using available data – HbA1C specially
- Enhanced monitoring with detailed ultrasound
- Multidisciplinary approach
Documentation and registry participation if available


What Guidelines Say?
Management Flow Chart
Emerging registry and cohort data suggest GLP-1 RAs are not associated with increased risk of major congenital malformations compared to insulin. However, pregnancy losses and terminations remain elevated in populations with obesity and T2DM, reflecting underlying maternal risk. The absence of randomized controlled trials, limited long-term developmental follow-up, and regional healthcare variations warrant caution.
Do Comparative Risk Assessment
CME INDIA Take Home
| GLP-1 RAs should not be initiated or continued in pregnancy. |
| Preconception counseling and planned transition to insulin are critical. |
| Inadvertent early pregnancy exposure appears not to increase major malformation risk, offering reassurance for affected patients. |
| Future research should focus on registries, mechanistic studies, and post-marketing surveillance to strengthen evidence. |
References:
- Huybrechts KF, Bateman BT, Hernández-Díaz S, et al. Safety of GLP-1 receptor agonists in early pregnancy: multinational cohort study. BMJ. 2024;385:e078852. doi:10.1136/bmj-2023-078852
- Jensen RB, Viuff AC, Pedersen LH, et al. Use of GLP-1 receptor agonists in pregnancy and reproductive safety: results from the European Teratology Information Services study. BMJ. 2024 Apr;385:e078699. doi:10.1136/bmj-2023-078699
- American Diabetes Association. 15. Management of diabetes in pregnancy: Standards of Care in Diabetes—2024. Diabetes Care. 2024;47(Suppl 1):S237–S248. doi:10.2337/dc24-S015
- European Medicines Agency. GLP-1 receptor agonists: product information and regulatory guidance. EMA; 2023. Available from: https://www.ema.europa.eu

Discover CME INDIA

- Explore CME INDIA Repository
- Examine CME INDIA Case Study
- Read History Today in Medicine
- Register for Future CMEs

