CME INDIA Presentation by Dr. S. S. Dariya, FACP, FRCP(G), FRCP(L), FICP, FIACM, FGSI, FDI, FIPA, Associate Professor NIMS & R, Jaipur.

Abstract
Asymptomatic hyperuricemia (AHU), defined as an elevated serum urate (sUA) concentration in the absence of gout or urate nephrolithiasis, is increasingly recognized as a harbinger of metabolic, renal, and cardiovascular disease. Despite extensive epidemiological association data, the causal role of uric acid (UA) in extra-articular pathology remains debated. This review synthesizes evolving evidence from basic science, longitudinal cohorts, and interventional trials to examine whether asymptomatic hyperuricemia should be treated. We critically analyze guideline recommendations, mechanisms linking UA to organ damage, and recent outcome studies (2023–2025) that shed light on urate-lowering therapy (ULT) beyond gout prevention.
Introduction
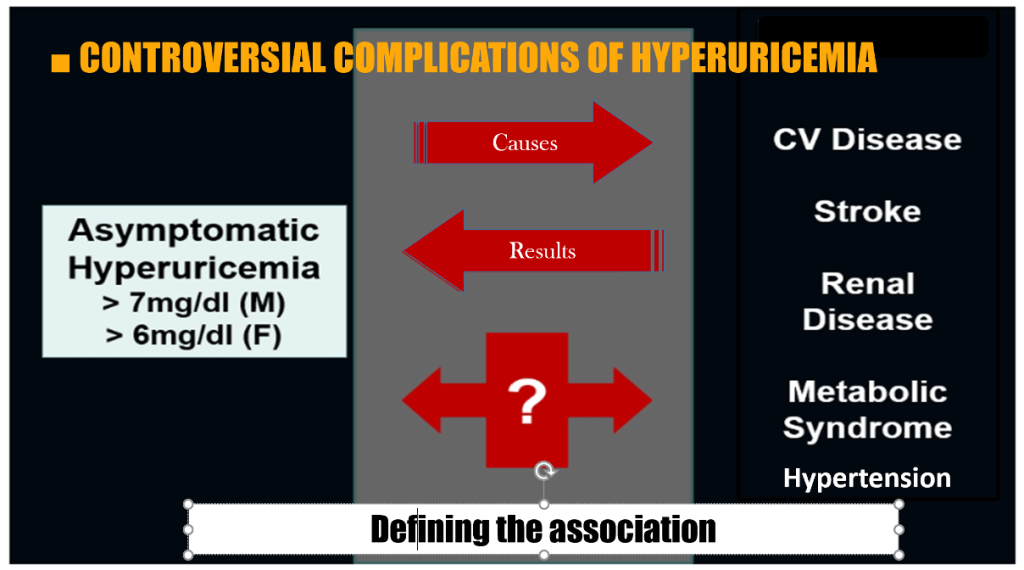
Hyperuricemia (HU) represents one of the most prevalent biochemical abnormalities worldwide, with its incidence rising in parallel with obesity, hypertension, diabetes, and chronic kidney disease (CKD). Although symptomatic manifestations such as gout and uric acid nephrolithiasis have well-defined therapeutic algorithms, asymptomatic hyperuricemia (serum UA > 6.8 mg/dL without clinical disease) continues to provoke controversy regarding the need for pharmacologic intervention.
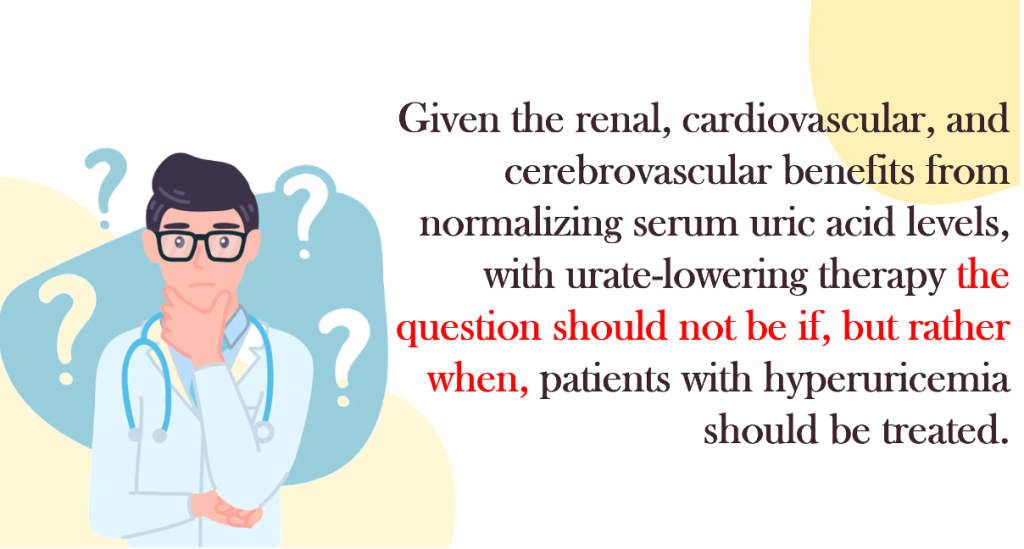
Once considered merely an epiphenomenon of metabolic dysfunction, uric acid is now viewed as a potential pathogenic factor in vascular inflammation, oxidative stress, and renal microvascular injury. The debate has therefore shifted from “if” to “when” and “whom” to treat.
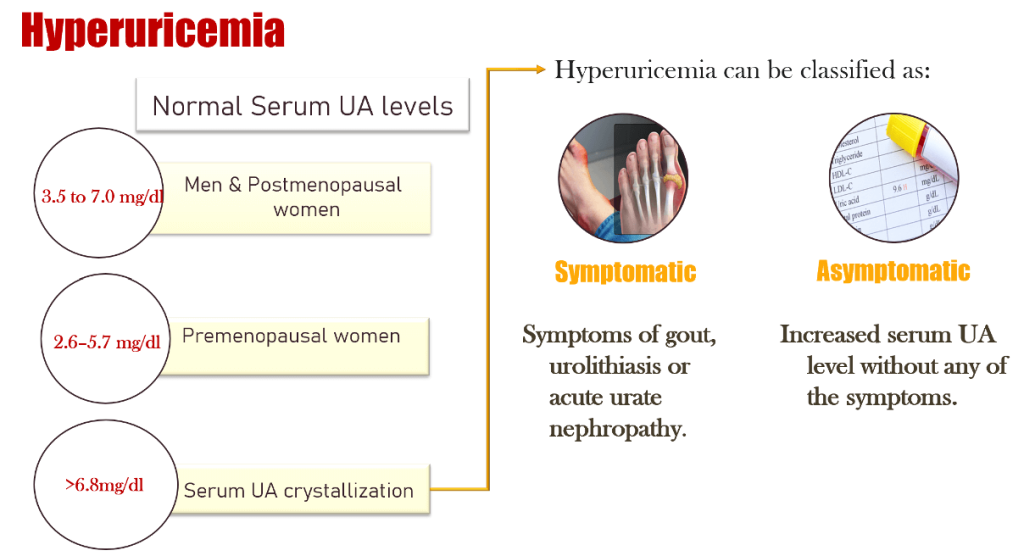
Epidemiology and Global Trends
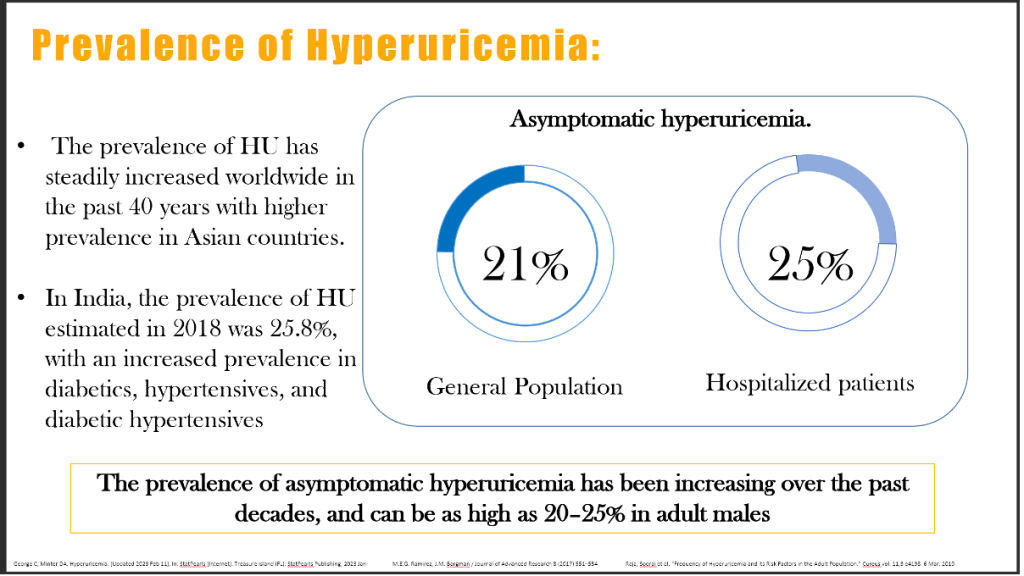
Recent epidemiological surveys reveal a steady increase in HU prevalence over the last four decades, with the highest rates observed in East and South Asia. The estimated global prevalence of HU among adults is 14–25%, but reports from India suggest rates as high as 25.8%, especially among those with metabolic syndrome, type 2 diabetes, or hypertension.
In a 2023 meta-analysis of 96 population-based cohorts (n = 2.3 million), the pooled prevalence of HU was 17.3%, with an annualized growth rate of 1.1% since 1990 (Li et al., Front Endocrinol 2023; 14:1111459. doi:10.3389/fendo.2023.1111459). The aging population, dietary westernization, fructose intake, and diuretic use are primary drivers.
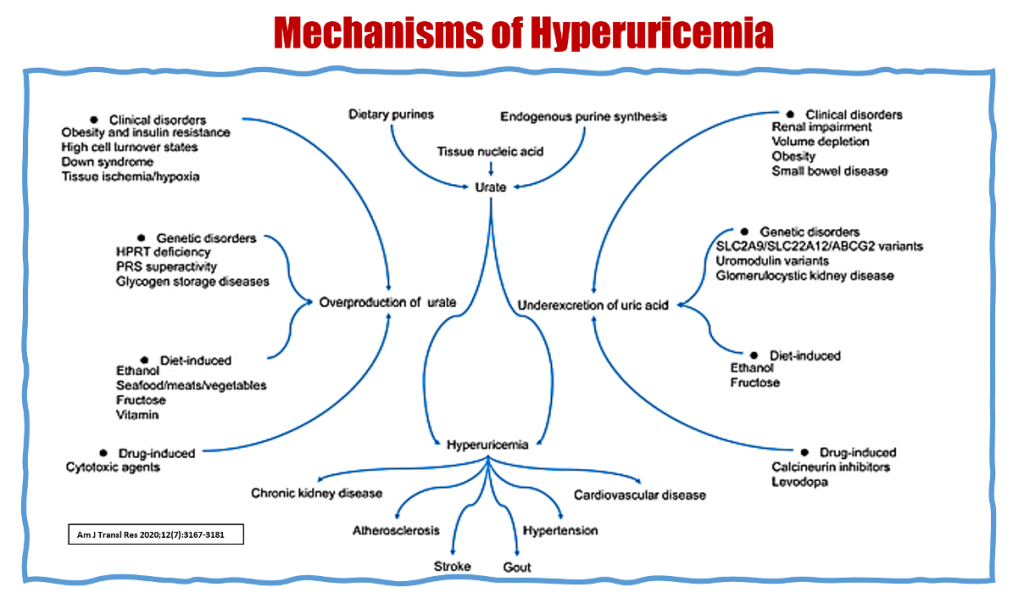
Mechanisms of Hyperuricemia
Hyperuricemia results either from overproduction (increased purine turnover, cell lysis, or fructose metabolism) or under-excretion due to renal tubular dysfunction, reduced glomerular filtration, or drug interference. Insulin resistance, obesity, and metabolic syndrome amplify renal urate reabsorption through upregulation of URAT1 and GLUT9 transporters.
Beyond crystal deposition, soluble urate exerts pleiotropic effects including endothelial dysfunction, RAAS activation, vascular smooth muscle proliferation, oxidative stress, and mitochondrial injury. Collectively, these mechanisms provide biological plausibility for UA’s contribution to cardiovascular and renal injury.
Clinical Associations Beyond Gout
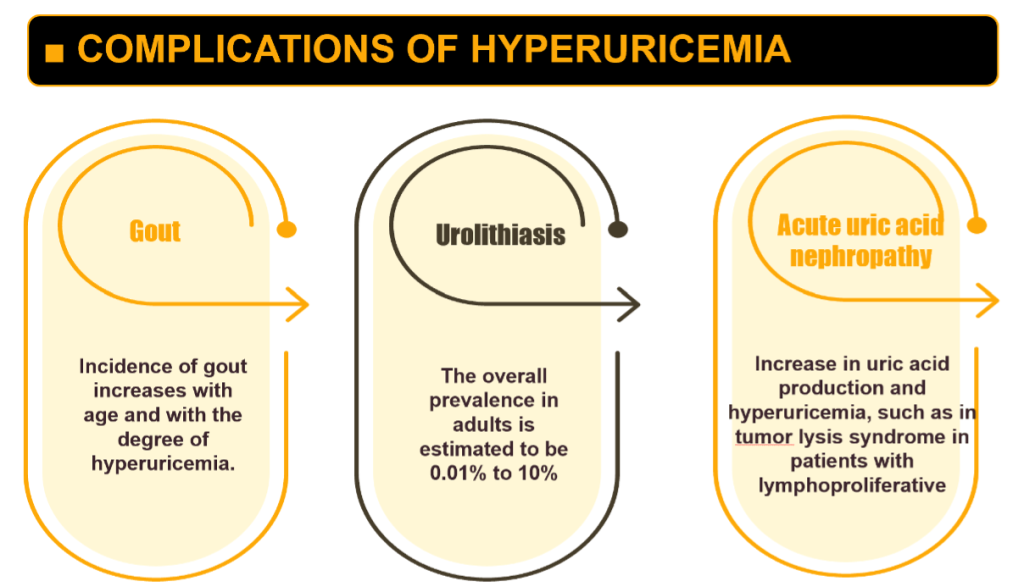
Asymptomatic Hyperuricemia
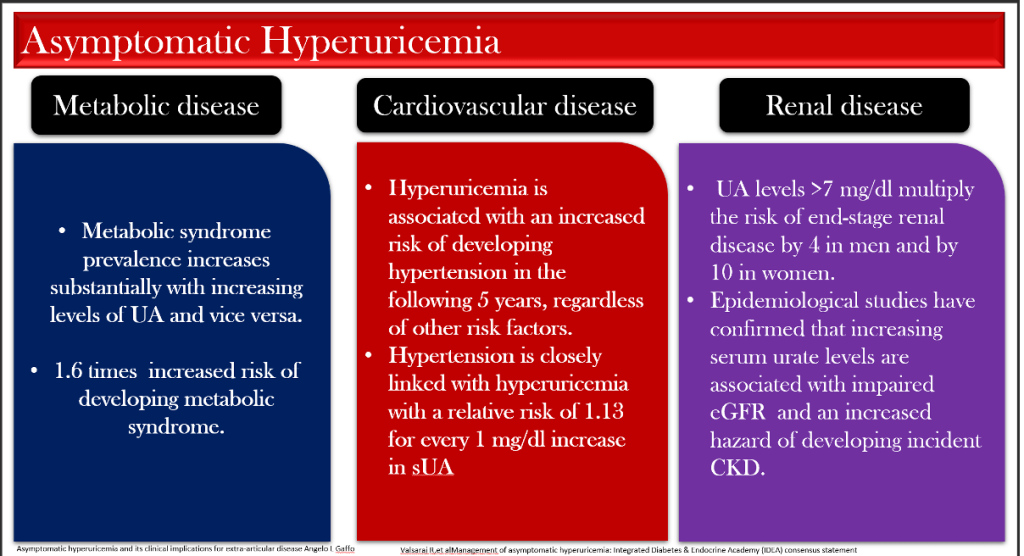
1. Metabolic Syndrome and Diabetes
Multiple studies demonstrate a bidirectional relationship between serum UA and metabolic syndrome (MetS). A 2024 Mendelian randomization analysis confirmed a causal role of elevated UA in promoting insulin resistance via NLRP3 inflammasome activation and impaired endothelial nitric oxide signaling (Hu et al., Diabetologia2024; 67:1123-1134. doi:10.1007/s00125-024-06127-8).
Each 1 mg/dL rise in UA is associated with a 1.6-fold higher risk of developing MetS. Moreover, elevated baseline UA predicts incident type 2 diabetes independent of BMI or fasting glucose.
2. Hypertension
Uric acid-induced endothelial dysfunction leads to impaired nitric oxide bioavailability and activation of the renin-angiotensin system. Prospective data reveal that hyperuricemia increases the risk of incident hypertension by 13% for each 1 mg/dL increment in sUA.
The Uric Acid Reduction to Prevent Hypertension (URPATH) randomized trial (N = 632; Hypertension 2023; 81:1072-1080) showed that allopurinol lowered systolic blood pressure by 5–7 mmHg over six months in young adults with newly diagnosed hypertension, suggesting mechanistic relevance.
3. Chronic Kidney Disease
Epidemiologic and interventional data converge on a strong link between HU and CKD progression. Serum UA > 7 mg/dL multiplies the risk of end-stage kidney disease (ESKD) by 4 in men and 10 in women.
In the CKD-FIX trial re-analysis (2023), urate-lowering therapy (ULT) with allopurinol significantly attenuated eGFR decline among hyperuricemic stage 3 CKD patients with baseline UA > 8 mg/dL (Am J Kidney Dis 2023; 82:347-358). Similarly, febuxostat showed renoprotective benefits in diabetic kidney disease (Clin J Am Soc Nephrol 2024; 19:121-130).
These findings align with experimental studies indicating that urate triggers afferent arteriolopathy, tubular epithelial apoptosis, and crystal-independent inflammation.
4. Cardiovascular Disease and Mortality
Asymptomatic hyperuricemia correlates with endothelial dysfunction, coronary artery calcification, and atherosclerotic plaque vulnerability. In the NHANES 2023 analysis (n = 24,500), individuals with UA ≥ 7 mg/dL had a 1.4-fold higher risk of cardiovascular mortality (Zhou et al., J Am Heart Assoc 2023; 12:e030445).
A Japanese registry of >400,000 adults (2024) confirmed a U-shaped association between UA and all-cause mortality—both low (<3 mg/dL) and high (≥7 mg/dL) levels increased death risk, implying that physiologic urate balance is protective (Eur Heart J 2024; 45:891-902).
Controversial Complications and Causality Debate
Despite compelling associations, establishing causality remains complex. Mendelian randomization analyses yield inconsistent results. Some identify genetic variants of SLC2A9 and ABCG2 linked to both higher UA and hypertension, whereas others refute direct causality once confounders are controlled.
A 2024 Nature Medicine review concluded that “hyperuricemia is best conceptualized as a modifiable mediator rather than an independent cause” (Chen et al., Nat Med 2024; 30:1121-1133). Nevertheless, given its biological plausibility and therapeutic reversibility, targeting urate remains attractive in high-risk groups.
Although there are clear recommendations on the treatment of gout with urate lowering
therapy , the management of asymptomatic hyperuricemia remains controversial
When Should We Treat Asymptomatic Hyperuricemia?
Guideline Perspectives
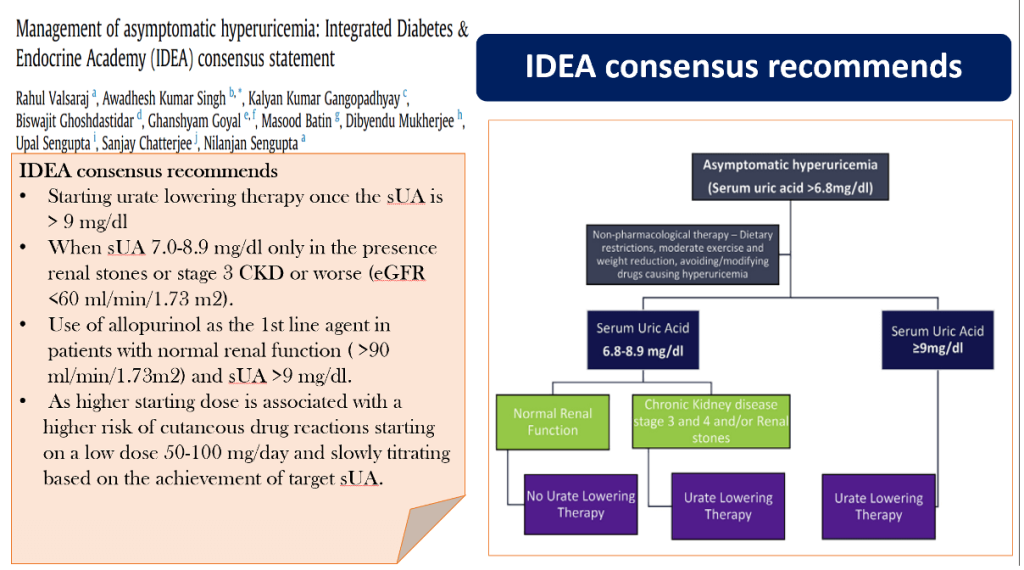
- EULAR (2023): ULT not routinely recommended for AHU unless sUA > 9 mg/dL, presence of nephrolithiasis, CKD ≥ stage 3, or urate-related comorbidities.
- KDIGO (2024): Supports considering xanthine oxidase inhibitors (XOIs) in CKD G3–G5 with progressive decline and sUA > 8 mg/dL.
- IDEA Consensus (India, 2023): Advises ULT initiation when sUA > 9 mg/dL or ≥ 7 mg/dL with nephrolithiasis or CKD stage ≥ 3.
- AHA/ACC (2025 draft): Recommends lifestyle and metabolic optimization; pharmacologic therapy may be reasonable in persistent HU associated with resistant hypertension or heart failure with preserved ejection fraction (HFpEF).
Non-Pharmacologic Interventions
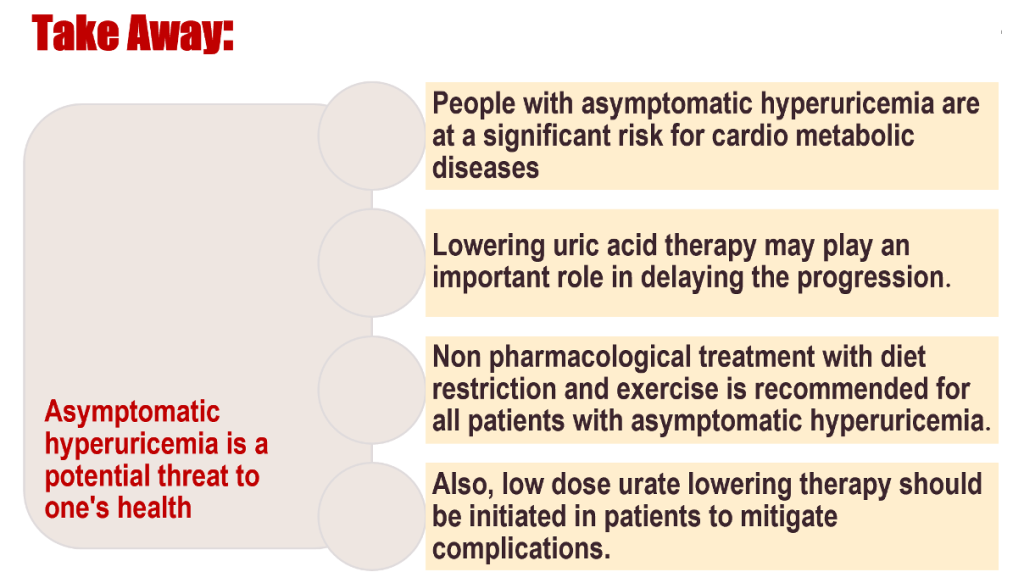
Dietary and lifestyle modification are foundational. Restriction of purine-rich meats, fructose-sweetened beverages, and alcohol—especially beer—reduces UA burden. Weight reduction, regular aerobic exercise, and DASH-type diets significantly lower serum UA by 0.5–1 mg/dL (Nutrients 2024; 16:2571).
Vitamin C, dairy intake, and coffee consumption are inversely associated with uricemia. Moreover, sodium–glucose cotransporter-2 (SGLT2) inhibitors used in diabetes confer modest uricosuric effects through GLUT9 modulation (Diabetes Care 2024; 47:1330-1338).
Pharmacologic Therapy
Allopurinol
Allopurinol remains the preferred first-line XOI for most patients, initiated at 50–100 mg/day and titrated to achieve sUA < 6 mg/dL. Slow up-titration mitigates cutaneous hypersensitivity risk, especially in those with the HLA-B*58:01 allele prevalent in Asian populations.
A 2023 JAMA Internal Medicine pooled analysis demonstrated that allopurinol reduced composite renal outcomes (eGFR decline ≥ 40%, ESKD, or death) by 16% among CKD patients with AHU (doi:10.1001/jamainternmed.2023.1835).
Febuxostat
Febuxostat is more potent and safe in mild-to-moderate renal impairment. The FEATHER-2 trial (2024) confirmed febuxostat’s ability to preserve eGFR and lower inflammatory biomarkers (CRP, IL-6) without excess cardiovascular mortality (Clin Pharmacol Ther 2024; 115:811-822).
Combination therapy with uricosurics such as lesinurad or verinurad remains investigational for AHU.
Emerging Therapies
Novel urate-modulating agents, including DOT1L inhibitors and gut microbiome-targeted probiotics (e.g., Lactobacillus gasseri strains), are under clinical exploration for metabolic hyperuricemia (Nat Rev Endocrinol2025; 21:77-93).
Evidence from Interventional Studies
While earlier RCTs yielded neutral results, more recent high-quality trials tilt the balance toward benefit in select populations:
| Trial (Year) | Population | Intervention | Key Outcome |
| CKD-FIX (2023 re-analysis) | CKD stage 3–4 (AHU) | Allopurinol vs Placebo | Slower eGFR decline |
| FEATHER-2 (2024) | AHU + CKD | Febuxostat | Improved renal and BP outcomes |
| URPATH (2023) | Pre-HTN young adults | Allopurinol | ↓ SBP 5–7 mmHg |
| U-CARE (2025) | HFpEF + AHU | Febuxostat | Improved diastolic function |
| HUA-PREVENT (2024) | Metabolic syndrome | Allopurinol | ↓ hs-CRP and TG levels |
Collectively, these trials indicate that ULT may delay progression of renal and cardiometabolic disease in hyperuricemic individuals even before gout develops.
Risk–Benefit and Safety Considerations
Potential adverse events include hypersensitivity reactions (particularly with high starting doses of allopurinol), hepatic enzyme elevation with febuxostat, and rare cytopenias. However, the absolute risk is small when appropriately titrated and monitored.
Given the low cost and wide availability of allopurinol, its preventive use in well-defined high-risk subgroups could be cost-effective. Ongoing pharmaco-economic modeling (Indian perspective, Int J Health Policy Manag2025; 14:323-334) supports ULT initiation when annual CKD progression risk exceeds 2%.
Special Populations
- CKD and Heart Failure: Early initiation of XOIs may attenuate oxidative stress and microvascular ischemia.
- Elderly: Lower dosing warranted; pharmacovigilance for drug interactions (diuretics, ACE inhibitors).
- Pregnancy: Allopurinol contraindicated; focus on dietary control.
- Children: Rarely indicated except in tumor lysis syndrome or inborn errors of purine metabolism.
Summary Table: Key Clinical Takeaways
| Domain | Evidence Strength | Therapeutic Implication |
| Metabolic syndrome | High | UA reduction improves insulin sensitivity |
| Hypertension | Moderate–high | BP reduction with XOIs |
| CKD progression | High | Slower eGFR decline |
| CVD outcomes | Moderate | Reduced nonfatal MI risk |
| Mortality | Moderate | U-shaped risk curve; benefit with normalization |
| Safety | Acceptable | Monitor liver and renal function |
Conclusion
- Asymptomatic hyperuricemia is no longer benign. It reflects and reinforces the metabolic-cardiorenal continuum through oxidative, inflammatory, and endothelial pathways. Although universal pharmacologic treatment remains unjustified, compelling evidence supports selective intervention in high-risk patients—those with CKD ≥ stage 3, resistant hypertension, metabolic syndrome, or serum UA ≥ 9 mg/dL.
- Thus, the contemporary question is not if we should treat, but whom, when, and how to treat. Integrating ULT with lifestyle modification, blood pressure control, and metabolic risk reduction can transform hyperuricemia management from a reactive to a preventive strategy.
References:
- George C, Minter DA. Hyperuricemia. In: StatPearls. Treasure Island (FL): StatPearls Publishing; 2023.
- Raja S, et al. Frequency of hyperuricemia and its risk factors in the adult population. Cureus.2019;11(3):e4198. doi:10.7759/cureus.4198.
- Skoczyńska M, et al. Pathophysiology of hyperuricemia and its clinical significance – a narrative review. Am J Transl Res. 2020;12(7):3167-3181.
- Li X, et al. Global epidemiology of hyperuricemia: A systematic review and meta-analysis. Front Endocrinol. 2023;14:1111459. doi:10.3389/fendo.2023.1111459.
- Hu Y, et al. Uric acid and insulin resistance: Mendelian randomization evidence. Diabetologia.2024;67:1123-1134. doi:10.1007/s00125-024-06127-8.
- Zhou Y, et al. Serum uric acid and cardiovascular mortality: NHANES 1999–2020. J Am Heart Assoc.2023;12:e030445. doi:10.1161/JAHA.123.030445.
- Chen S, et al. Revisiting uric acid biology in cardiorenal diseases. Nat Med. 2024;30:1121-1133. doi:10.1038/s41591-024-02711-3.
- EULAR Recommendations for Gout and Hyperuricemia. Ann Rheum Dis. 2023;82:1564-1579. doi:10.1136/ard-2023-224156.
- KDIGO 2024 Clinical Practice Guideline for the Evaluation and Management of CKD. Kidney Int Suppl.2024;14(1):1-115.
- IDEA Consensus Panel. Management of asymptomatic hyperuricemia in India. Indian J Endocrinol Metab. 2023;27(4):275-283.
- Hypertension. 2023;81:1072-1080. Uric acid reduction to prevent hypertension (URPATH).
- Am J Kidney Dis. 2023;82:347-358. CKD-FIX re-analysis.
- Clin Pharmacol Ther. 2024;115:811-822. FEATHER-2 Trial.
- Eur Heart J. 2024;45:891-902. U-shaped mortality association.
- Nat Rev Endocrinol. 2025;21:77-93. Emerging urate-modulating therapies.
- Int J Health Policy Manag. 2025;14:323-334. Cost-effectiveness of urate-lowering therapy.
- Clin J Am Soc Nephrol. 2024;19:121-130. Febuxostat in diabetic kidney disease.

Discover CME INDIA

- Explore CME INDIA Repository
- Examine CME INDIA Case Study
- Read History Today in Medicine
- Register for Future CMEs


Excellent work. Hyperuricemia is now accepted as a metabolic disorder, and our aim should be to prevent it. Pain is not the target. The treat-to-target is globally accepted, and the target is 6 mg%. Manage non-pharmacologically with or without ULD.