CME INDIA Case Presentation by Dr. Kirti Soota, Consulting Diabetes Physician, Delhi; Dr. Brij Makkar, Past President, RSSDI, Senior Diabetes & Bariatric Physician, Director – Dr. Makkar’s Diabetes and Obesity Centre, Delhi. X Handles: @KirtiSoota @DrBMMakkar
CME INDIA Case Study

Abstract
This case report details the diagnosis and management of a 16-year-old lean female with a 6-year history of diabetes, initially controlled with diet and later adequately managed with vildagliptin and metformin. The patient’s strong multigenerational family history, preserved C-peptide levels, negative autoimmune markers, and a confirmed HNF1A gene mutation (c.485T>C; p.Leu162Pro) led to a diagnosis of Maturity-Onset Diabetes of the Young Type 3 (MODY 3). Notably, the patient has shown an unexpected positive response to DPP-4 inhibitor and metformin therapy, challenging the conventional sulfonylurea-centric approach for HNF1A-MODY. This case underscores the importance of genetic testing in young, non-obese diabetic patients and highlights the need for personalized management strategies.
Introduction
Maturity-Onset Diabetes of the Young (MODY) is a monogenic form of diabetes characterized by autosomal dominant inheritance, early onset (typically before age 25), and preservation of β-cell function. Accounting for 1-5% of all diabetes cases, MODY is often misdiagnosed as Type 1 or Type 2 diabetes due to overlapping clinical features. HNF1A-MODY (MODY 3) is the most common subtype, caused by mutations in the HNF1A gene, and is known for its sensitivity to sulfonylureas (SUs). This case presents a unique scenario where a young patient with genetically confirmed MODY 3 responds well to a DPP-4 inhibitor and metformin, prompting a re-evaluation of therapeutic guidelines.
Case Presentation
Patient Details : Age/Sex: 16-year-old female
Anthropometry: Height 147 cm, Weight 48 kg, BMI ~22 kg/m² (lean build), Waist Circumference 70 cm
Clinical History: Diagnosed with diabetes at age 10 (initial HbA1c 6.8%), managed with diet for 6 years.
Recent HbA1c 8.1% , started on vildagliptin (50 mg) + metformin (500 mg twice daily).
No obesity, insulin resistance, or metabolic syndrome; normal lipid profile and liver function tests.
Family History : Strong multigenerational diabetes history: father, paternal grandmother, paternal uncle, and maternal grandfather.
Suggests autosomal dominant inheritance, a hallmark of MODY.
Investigations
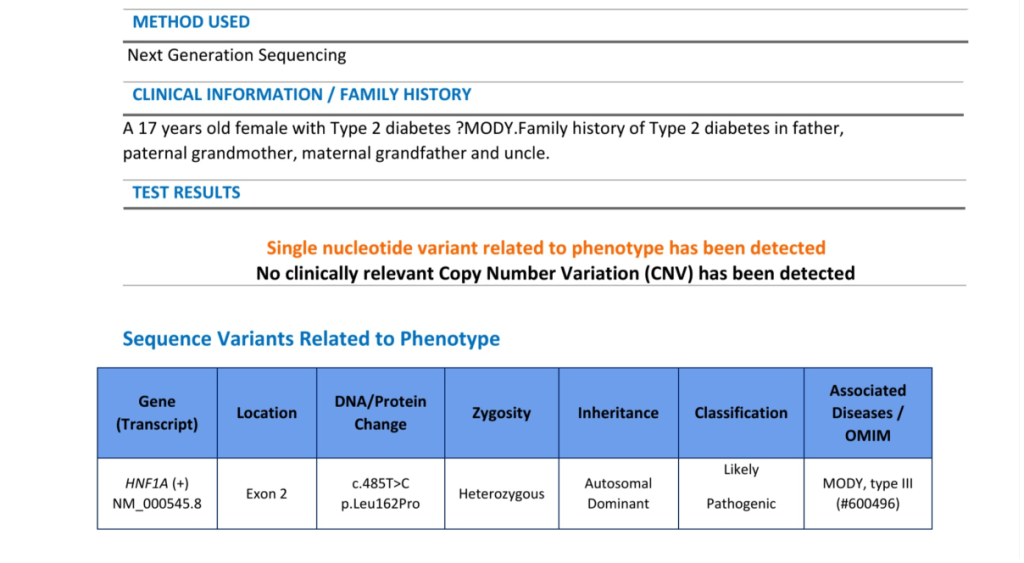
Genetic Testing (Next-Generation Sequencing – MODY Panel): Variant Identified: HNF1A c.485T>C; p.Leu162Pro (heterozygous, exon 2).
Classification: Likely Pathogenic (ClinVar, ACMG criteria).
Disease Association: MODY Type 3 (OMIM #600496).
Inheritance: Autosomal dominant.
Biochemical Markers:
C-Peptide: 2.73 ng/mL (normal range 1.10–4.40 ng/mL), indicating preserved β-cell function.
Autoimmune Profile (Type 1 Diabetes Antibodies): GAD-65: <10 IU/mL (negative).
| IA-2: <10 IU/mL (negative). |
| ZnT8: <15 U/mL (negative). |
| Islet Cell Ab: Negative. |
| Insulin Ab: 6.45 µU/mL (within reference range). |
| Overall: Negative autoimmune panel, ruling out Type 1 diabetes or latent autoimmune diabetes in adults (LADA). |
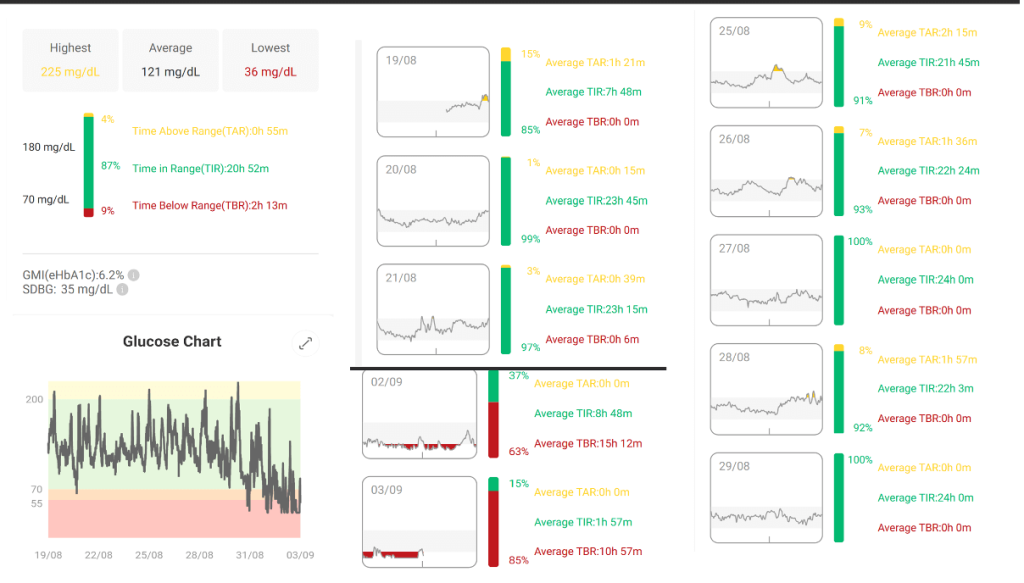
Glucose Monitoring (Continuous Glucose Monitoring – CGM Data): Average glucose: 121 mg/dL.
Time in Range (TIR, 70-180 mg/dL): 87%.
Time Above Range (TAR, >180 mg/dL): 4%.
Time Below Range (TBR, <70 mg/dL): 9%.
Estimated HbA1c (eA1c): 6.2%.
Standard Deviation (SDBG): 35 mg/dL.
CGM trends show stable glycemic control with occasional hypoglycemia, likely due to metformin.
Interpretation & Diagnosis
- The diagnosis of MODY 3 (HNF1A mutation) is supported by: Early onset at age 10.
- Strong autosomal dominant family history.
- Lean build with no metabolic syndrome.
- Preserved C-peptide and negative autoantibodies.
- Confirmed pathogenic HNF1A mutation.
Discussion
Diagnostic Challenges
MODY is frequently misdiagnosed due to its phenotypic similarity to Type 1 and Type 2 diabetes. The patient’s young age mimicked Type 1 diabetes, while the family history suggested Type 2 diabetes. However, the preserved C-peptide and negative autoimmune markers prompted genetic testing, confirming MODY 3.
The HNF1A c.485T>C mutation is a well-documented variant associated with MODY 3, affecting hepatocyte nuclear factor-1α, a transcription factor critical for β-cell function (Ellard et al., 2020).
Unexpected Therapeutic Response
Conventionally, HNF1A-MODY is highly sensitive to sulfonylureas due to the β-cell’s retained ability to secrete insulin in response to glucose. Studies recommend low-dose glimepiride or gliclazide as first-line therapy (Shepherd et al., 2016). However, this patient’s HbA1c improved from 8.1% to an estimated 6.2% (via CGM) on vildagliptin and metformin, suggesting a partial response to DPP-4 inhibition. This is unusual, as DPP-4 inhibitors are less effective in MODY 3 compared to SUs, likely due to the mechanism of incretin enhancement versus direct insulin secretion stimulation (Pearson et al., 2003).
The metformin contribution may stabilize hepatic glucose output, complementing the DPP-4 effect. This response warrants further investigation into individual variability in MODY 3 treatment.
Management Implications
Therapeutic Adjustment: A trial of low-dose sulfonylurea (e.g., gliclazide 20-40 mg/day) is recommended to optimize glycemic control, given the genetic diagnosis. Vildagliptin and metformin can be tapered if SU proves effective.
Family Screening: Cascade genetic testing of affected relatives is crucial to identify other MODY cases.
Long-Term Monitoring: Regular screening for microvascular complications (retinopathy, nephropathy, neuropathy) is essential, as MODY 3 patients are at risk despite good control.
Genetic Counselling: Education on autosomal dominant inheritance and long-term prognosis is advised.
Clinical Relevance
- This case highlights the importance of genetic testing in young, non-obese diabetic patients with a family history. The unexpected response to DPP-4 inhibitor and metformin suggests that treatment guidelines may need refinement to account for individual variability.
- Future research should explore the molecular basis of this response, potentially involving incretin pathway modulation in HNF1A-MODY.
Conclusion
This 16-year-old female with genetically confirmed MODY 3 presents a classic case of monogenic diabetes, supported by clinical, biochemical, and genetic evidence. Her positive response to vildagliptin and metformin challenges the sulfonylurea-centric paradigm, offering insights into personalized diabetes management.
Early diagnosis through genetic testing and tailored therapy can significantly improve outcomes in MODY patients.
References:
- Ellard, S., et al. (2020). “Best practice guidelines for the molecular genetic diagnosis of maturity-onset diabetes of the young.” Diabetologia, 63(10), 2000-2011.
- Pearson, E. R., et al. (2003). “Switching from insulin to oral sulfonylureas in patients with diabetes due to Kir6.2 mutations.” New England Journal of Medicine, 349(16), 1551-1559.
- Shepherd, M., et al. (2016). “Systematic population screening, using biomarkers and genetic testing, identifies 2.5% of the UK pediatric diabetes population with monogenic diabetes.” Diabetes Care, 39(11), 1879-1888.
Discover CME INDIA:

- Explore CME INDIA Repository
- Examine CME INDIA Case Study
- Read History Today in Medicine
- Register for Future CMEs