CME INDIA Presentation by Dr. Kannan Natarajan, MBBS, DTCD, PG Dip.Diab, M.Diab, FCCP, Fellow Diabetes India, FRSSDI, Consultant Chest Physician and Diabetologist, Chandra Chest and Diabetes Care Centre, Chennai.
(Based on a presentation at CG-RSSDI 2024,Raipur on 21st December)
It all starts with:
- Patients with diabetes are over-represented among the total cases reported with “idiopathic” pulmonary fibrosis (IPF).
- This raises the question, whether this is an association only or whether diabetes itself can cause pulmonary fibrosis.
- The lung is a target organ for diabetic microangiopathy, in both type I and type II diabetes.
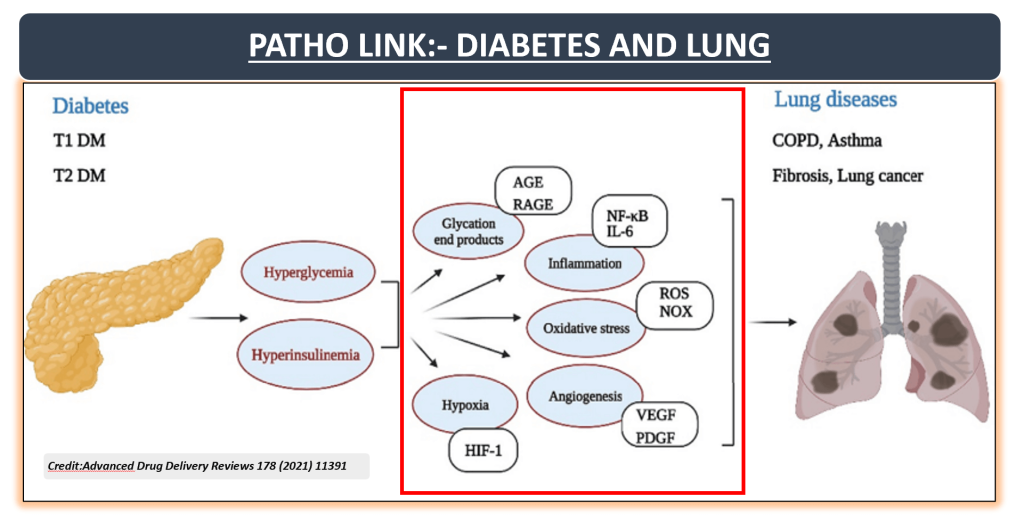
Evidence from studies highlights the strong link between lung diseases and diabetes
| Incidence |
| COPD: Individuals with diabetes have a 22-33% higher prevalence of COPD. |
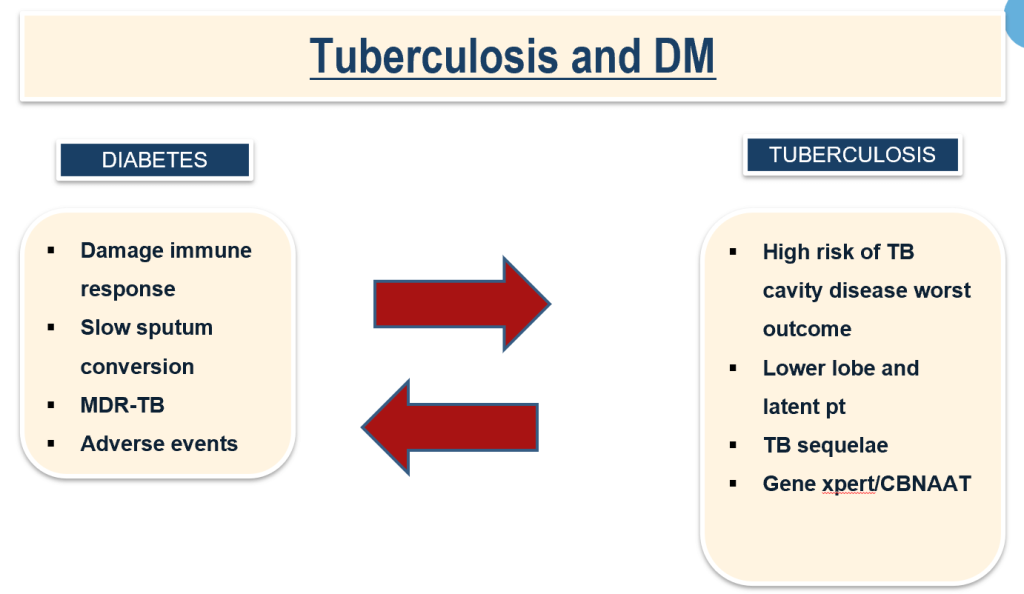
| TUBERCULOSIS:4 FOLDS IN DM, 30% OF TB WILL HAVE DM. |
| Asthma: Around 10-12% of individuals with asthma also have diabetes, with worsened symptoms. |
| Pulmonary Fibrosis (IPF): Diabetes increases the risk of idiopathic pulmonary fibrosis and worsens lung function. |
| Pneumonia: Diabetic individuals are 3-4 times more likely to develop pneumonia. |
| Sleep Apnea: 50-80% of people with type 2 diabetes may suffer from obstructive sleep apnea. |
Structural changes in lung infection patient with DM
| Narrowing of the alveolar space. |
| Flattening of the alveolar. |
| Expansion of the interstitium. |
| Involvement of the pulmonary vessels. |
| Involvement of the BM of the alveolar epithelium. |
| Bronchial epithelium and the pulmonary capillaries. |
Respiratory abnormalities and DM
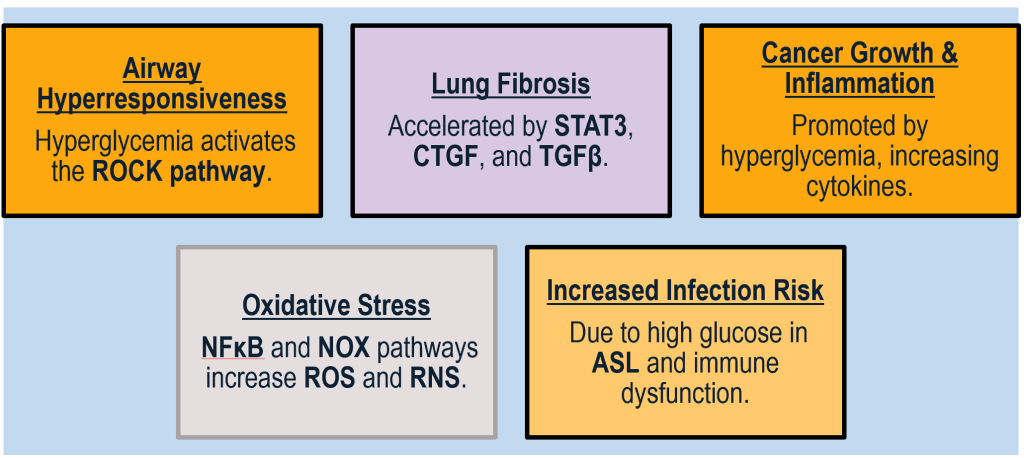
The downstream effects of hyperglycemia on various biological processes.
| Hyperglycemia: which lead to multiple complications. |
| Rho/Rock: This pathway can lead to: |
| Airway hyperresponsiveness: Increased sensitivity of the airways, which can result in breathing difficulties. |
| STAT3, CTGF, TGF β: These growth factors and signal transducers are associated with: |
| Fibrosis: The thickening and scarring of connective tissue as a result of chronic inflammation. |
| ROS (Reactive Oxygen Species), RNS (Reactive Nitrogen Species), NOX (NADPH oxidase): These are linked to several pathways: |
| Malignancy: Increased risk of cancer due to oxidative damage and inflammation. |
| Oxidative stress: Cellular damage caused by an imbalance between antioxidants and free radicals. |
| Inflammation: General body response to harmful stimuli, involving immune cells, blood vessels, and molecular mediators. |
| NF-kB: Nuclear factor kappa-light-chain-enhancer of activated B cells, which is involved in: |
| Inflammatory cytokine release: The production and release of cytokines that promote inflammation. |
| ASL (Airway Surface Liquid), hyperglycemia: This condition can lead to: |
| Infection: Increased susceptibility to infections due to changes in the environment of the respiratory system. |
Insulin’s effects on the lung
- Inhibition of Surfactant Proteins: Insulin inhibits the production of surfactant protein A (SP-A) and surfactant protein D (SP-D) through the phosphoinositide-3–kinases (PI3K) pathway. These proteins are crucial for lung surfactant function, which reduces surface tension in the alveoli, aiding in lung expansion.
- Immune Response Modulation: Insulin inhibits the proliferation of T helper cell type 1 (Th1) cells, which are crucial in cell-mediated immunity and response to viral infections. It shifts the immune balance towards T helper cell type 2 (Th2) responses, which are more involved in antibody production and allergic reactions.
- Mast Cell Activity: Insulin promotes mast cell survival, degranulation, and histamine release, which are key in allergic responses and inflammation. This effect is mediated through the PI3K pathway.
- Airway Smooth Muscle (ASM) Activity: Insulin influences airway smooth muscle cells (ASMs) by promoting their proliferation and contraction through pathways involving mitogen-activated protein kinase (MAPK), Rho kinase, and PI3K. This can contribute to airway hyper-responsiveness and narrowing, characteristic of asthma and COPD.
- Fibrosis and ECM Deposition: Insulin increases the deposition of extracellular matrix (ECM) in the lung, which can lead to stiffening and scarring of lung tissue. It promotes epithelial-mesenchymal transition (EMT) and fibrosis through the activation of the PI3K/protein kinase B β-catenin pathway. This transition can lead to more fibrotic tissue, reducing lung elasticity and function.
- Inflammatory Response: Insulin activates pulmonary inflammatory macrophages, which play a role in the lung’s inflammatory response, potentially exacerbating conditions like chronic inflammation or acute respiratory distress syndrome (ARDS).
Each of these points highlights how insulin can modulate lung physiology and pathology, influencing immune responses, cellular function, and tissue remodelling, which are critical in the context of respiratory diseases.
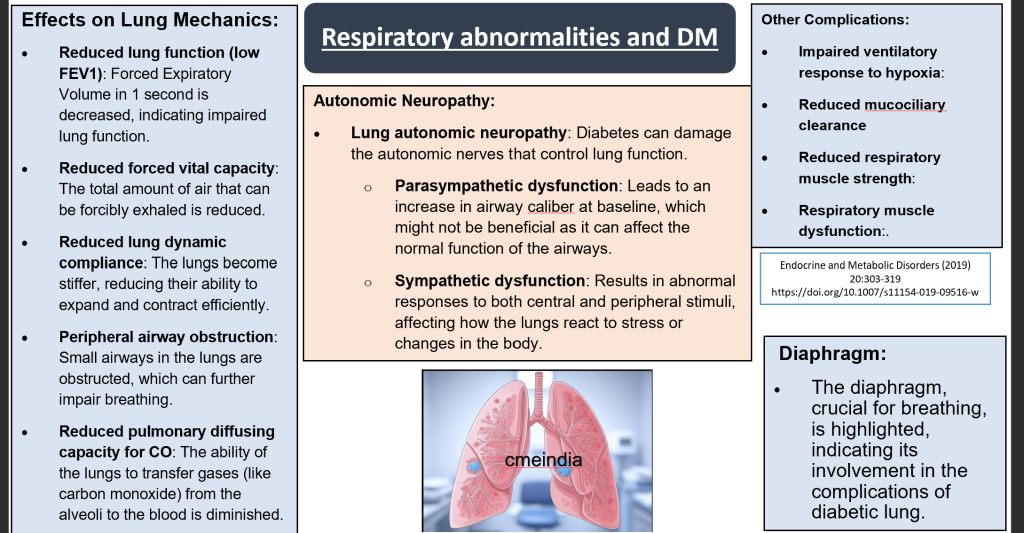
The first identified test -Pulmonary function test
- Common simple lung function tests alone are likely to underestimate the prevalence and degree of lung dysfunction in diabetes mellitus patients.
- FEV1/FVC are significantly lower in PWD than controls, Average rate of decline in FEV1 is 71 ml/year in diabetes patients.
- An increase in HbAlc is associated with decrease in lung function parameters FEV1 and FVC (impaired glucose auto-regulation is associated with impaired lung function).
Pulmonary complications of diabetes
| Increased risk of pneumonia and lung infections in diabetics. |
| Chronic obstructive pulmonary disease (COPD)/Asthma exacerbated by diabetes. |
| Pulmonary fibrosis due to impaired DNA repair and inflammation. |
| Reduced lung function from diabetes-induced inflammation and fibrosis. |
| Diabetic pneumopathy with poor lung tissue repair and . |

The relationship between diabetes mellitus (DM) and lung cancer
Lung cancer and DM
- Increased Risk: Diabetes is linked to a higher risk of developing lung cancer.
- Chronic Inflammation: Inflammation in diabetes may promote lung cancer progression.
- Insulin Resistance: Insulin resistance in diabetes can fuel tumor growth in the lungs.
- Worse Prognosis: Diabetic patients with lung cancer tend to have poorer outcomes.
- Higher Incidence: Type 2 diabetes increases the likelihood of lung cancer occurrence.

DM and Lung Infection
- Immune System Impairment: Diabetes mellitus (DM) compromises various immune functions, including neutrophil and macrophage activities crucial for fighting infections, due to factors like hyperglycemia and insulin resistance. This leads to a higher susceptibility to infections.
- Increased Susceptibility: Diabetic patients are more prone to lung infections due to altered immune responses and higher glucose levels in the lungs, which can promote bacterial overgrowth. Common pathogens include Staphylococcus aureus, Streptococcus pneumoniae, influenza virus, Klebsiella pneumoniae, Pseudomonas aeruginosa, and fungi like Mucorales and Aspergillus species.
- Hospitalization Risks: During influenza epidemics, hospitalization rates for diabetic patients are significantly higher than for non-diabetic individuals. Poor glycemic control in DM, indicated by elevated HbA1c levels, correlates with increased pneumonia severity, hospitalization risks, and mortality.
- Prognosis and Treatment: DM is linked with a worse prognosis in community-acquired pneumonia, with higher rates of complications like pleural effusion. Early antibiotic treatment within 8 hours of initial assessment can reduce complications and mortality in diabetic patients with pneumonia.
- Preventive Measures: While vaccination against pneumococcal infections is recommended, its effectiveness in diabetic patients might be reduced, possibly due to lower vaccine uptake or efficacy. Medications like angiotensin-converting-enzyme inhibitors and statins might offer some protection against pneumonia in diabetic patients, though the mechanisms are not fully understood.
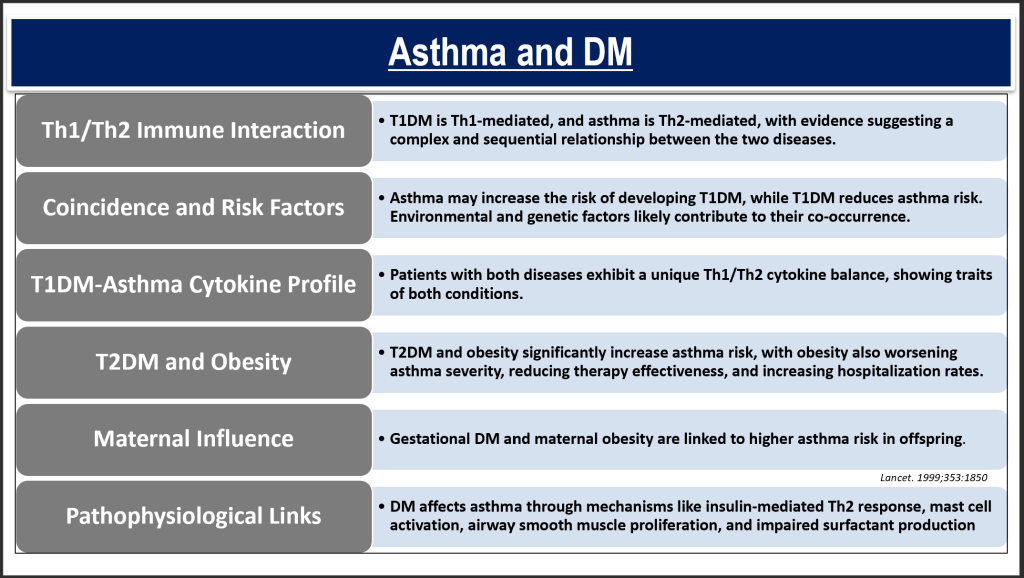
Type 2 Diabetes Mellitus (T2DM) and asthma
- Its prevalence show inconsistency, likely due to variations in study methodologies and participant demographics.
- Nonetheless, research indicates that T2DM and obesity could exacerbate asthma risk, with obesity itself linked to a reduced effectiveness of asthma treatment and a higher hospitalization rate for asthma exacerbations, around five times more than in non-obese asthmatics.
- Furthermore, conditions such as gestational diabetes and maternal obesity are associated with a heightened risk of asthma.
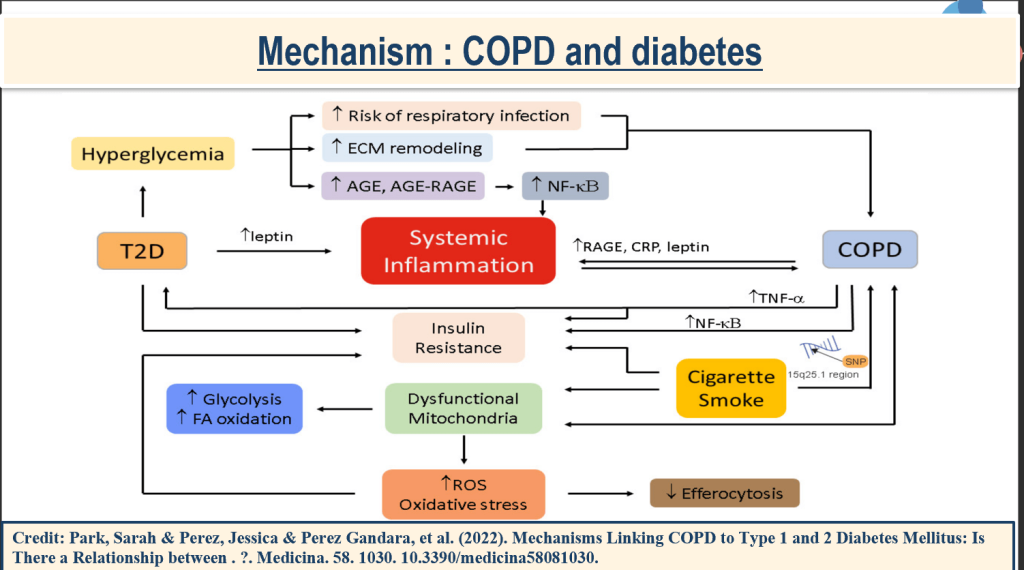
Diabetes & COPD

Idiopathic pulmonary fibrosis and DM
- Idiopathic pulmonary fibrosis (IPF) is a chronic, progressive, fibrosing interstitial pneumonitis of by complex pathophysiology, with O2 dependence.
- Affects Older Adults: Primarily occurs in older adults/ corpulmonale/PAH.
- Key Features: Characterized by progressive worsening of dyspnea and lung function.
- Prognosis: Associated with a poor prognosis/honey combing, groundglass, right heart failure.
- No Extrapulmonary Manifestations: No significant extrapulmonary manifestations have been recognized; it is assumed to be limited to the lung.

Pulmonary hypertension and DM
- Pulmonary Hypertension (PH): Elevated pulmonary pressure causing right heart failure and death.
- Diabetes Impact: Diabetes may contribute to PH through vascular damage, fibrosis of right heart via platelet derived growth factor (pdgf).
- Higher Incidence: Diabetes is more common in PH patients than the general population.
- Unclear Significance: Clinical relevance remains uncertain due to pulmonary capillary reserve.

Diabetes and Cystic Fibrosis (CFRD)

Metformin and Lung Diseases
- Metformin, an oral antidiabetic drug, has been extensively studied for its potential benefits beyond diabetes management, particularly in lung diseases due to its antidiabetic, antioxidant, and anti-inflammatory properties.
- Lung Cancer: Metformin’s role in lung cancer continues to be debated. While some studies suggest improved survival outcomes in lung cancer patients using metformin, particularly in non-small cell lung cancer, others find no significant anti-tumor efficacy in lung cancer, unlike in other cancers. This discrepancy indicates that the protective or therapeutic effects of metformin on lung cancer are not yet definitively established.
- Lung Infections: Metformin has demonstrated the ability to enhance macrophage bactericidal activity, potentially offering protection
Insulin and Lung Health:
- Oncogenic Effects: Insulin has been linked to non-small cell lung cancer due to its oncogenic activity. This is in contrast to metformin, which has potential therapeutic benefits in lung diseases. Insulin-like growth factor receptor 1 (IGF-1) is also associated with lung cancer development and resistance to chemotherapy, indicating a complex role of insulin in cancer progression.
- Impact on Respiratory Function: Insulin influences various cellular components in the lung, potentially leading to airway hyper-responsiveness, bronchoconstriction, and increased inflammation. This suggests insulin may exacerbate conditions like asthma. Studies have indicated that insulin might shift T-cell differentiation towards a Th2 response, which is implicated in asthma pathogenesis. A Taiwanese study highlighted that insulin use could increase asthma risk.
PPAR-γ Agonists and Lung Health:
- Anti-inflammatory Effects: PPAR-γ agonists like pioglitazone and rosiglitazone have been studied for their potential to reduce lung inflammation. They decrease the influx of neutrophils into alveolar spaces, thus reducing lung injury and inflammation in both in vitro studies and animal models. These agonists also inhibit the production of pro-inflammatory cytokines, which is beneficial in conditions like ischemia reperfusion injury.
- Anti-fibrotic Properties: Pioglitazone shows promise in managing fibrotic lung conditions, such as idiopathic pulmonary fibrosis (IPF) and bleomycin-induced lung injury, by reducing fibrotic processes in the lung tissue.
- Asthma Management: While rosiglitazone showed benefits in animal models of asthma by improving lung function and reducing airway inflammation, these effects were not replicated in human clinical trials.
- Anti-cancer Potential: PPAR-γ agonists exhibit anti-tumor and anti-proliferative properties, suggesting their potential as therapeutic agents in lung cancer, although more research is needed.
Insulin secretagogues like sulfonylureas,& DPP4-Inhibitors
- Effects on Allergic Inflammation: Research has indicated that DPP-4 inhibitors might influence allergic airway inflammation and could regulate immunological pathways in asthma through CD26 interaction. However, clinical studies have not found a direct link between the use of DPP-4 inhibitors and asthma control, suggesting that while these drugs may have immunomodulatory effects, they do not significantly alter asthma outcomes in practice.
- Cancer Risk: There has been some concern regarding the potential oncogenic effects of insulin secretagogues like sulfonylureas, with earlier studies suggesting an increased risk of overall cancer. However, more recent meta-analyses have not found a significant association between sulfonylurea use and lung cancer risk specifically.
- Potential in Cancer Treatment: Some studies propose that sulfonylureas could be used in combination with chemotherapy to target resistant lung cancer cells, indicating a potential dual role in cancer therapy and glucose management.
Glucagon-like Peptide 1 Agonists (GLP-1) and Lung Health:
- Presence in Lung Tissue: The GLP-1 receptor is present in human lung tissue, which implies that the lungs could potentially be influenced by GLP-1 agonists, a class of drugs primarily used for managing type 2 diabetes and obesity.
- Potential Therapeutic Effects: Studies on animal models have suggested that GLP-1 agonists might play a role in treating obstructive pulmonary diseases. They have been shown to reduce the severity of acute exacerbations and decrease mortality in models of obstructive lung disease, possibly through mechanisms involving the enhancement of surfactant production, which is crucial for lung function.
- Anti-inflammatory Actions: There’s an indication that GLP-1 may exert anti-inflammatory effects in the lungs by downregulating pro-inflammatory cytokines like TNFα and NF-κB, which are key players in the inflammatory response.
- Cell Proliferation and Migration.
Sodium-Glucose Cotransporter 2 (SGLT2) Inhibitors and Lung Health:
- Primary Use: SGLT2 inhibitors are primarily used as oral medications for managing type 2 diabetes by reducing glucose reabsorption in the kidneys, which helps lower blood sugar levels.
- Cardiovascular Benefits: Beyond glucose control, these drugs have shown significant benefits in reducing cardiovascular risks, including heart failure hospitalizations and cardiovascular death, in diabetic patients.
- Potential Pulmonary Effects: While their cardiovascular impacts are well-documented, their effects on lung health are less explored. A recent in vitro study has hinted at a potential role in pulmonary health by suggesting that SGLT2 might contribute to the relaxation of human pulmonary artery smooth muscle cells through mechanisms involving nitric oxide (NO), which could theoretically benefit conditions involving pulmonary hypertension or other forms of pulmonary vascular disease.

CME INDIA Take-home message
- Diabetes impacts multiple organs, including the lungs, but pulmonary complications remain poorly studied compared to other diabetes-related comorbidities.
- The mechanisms of diabetes-induced lung disorders are not well understood, necessitating detailed mechanistic research.
- Managing pulmonary risks requires targeting multiple diabetes-related factors beyond glycemic control.
- The effects of insulin and anti-diabetic drugs on lung health need further exploration to clarify their direct roles.
- Experimental and large-scale human studies are essential for advancing understanding and management of diabetic lung disease.
- Early detection and treatment of lung complications in diabetic patients can improve quality of life and survival, highlighting public health importance.
References:
- Kolahian S, Leiss V, Nürnberg B. Diabetic lung disease: fact or fiction? Rev Endocr Metab Disord. 2019 Sep;20(3):303-319. doi: 10.1007/s11154-019-09516-w.
- Uppal P, Mohammed SA, Rajashekar S, Giri Ravindran S, Kakarla M, Ausaja Gambo M, Yousri Salama M, Haidar Ismail N, Tavalla P, Hamid P. Type 2 Diabetes Mellitus and Asthma: Pathomechanisms of Their Association and Clinical Implications. Cureus. 2023 Mar 12;15(3):e36047. doi: 10.7759/cureus.36047. PMID: 37056543; PMCID: PMC10089620.
- Park, Sarah & Perez, Jessica & Perez Gandara, Brais & Agudelo, Christina & Rodriguez Ortega, Romy & Ahmed, Huma & Garcia-Arcos, Itsaso & McCarthy, Cormac & Geraghty, Patrick. (2022). Mechanisms Linking COPD to Type 1 and 2 Diabetes Mellitus: Is There a Relationship between Diabetes and COPD?. Medicina. 58. 1030. 10.3390/medicina58081030.
- Khateeb J, Fuchs E, Khamaisi M. Diabetes and Lung Disease: A Neglected Relationship. Rev Diabet Stud. 2019 Feb 25;15:1-15. doi: 10.1900/RDS.2019.15.1. PMID: 30489598; PMCID: PMC6760893.
- un YH, He L, Yan MY, Zhao RQ, Li B, Wang F, Yang Y, Yu HP. Overexpression of GLP-1 receptors suppresses proliferation and cytokine release by airway smooth muscle cells of patients with chronic obstructive pulmonary disease via activation of ABCA1. Mol Med Rep. 2017;16(1):929–936. doi: 10.3892/mmr.2017.6618.

Discover CME INDIA

- Explore CME INDIA Repository
- Examine CME INDIA Case Study
- Read History Today in Medicine
- Register for Future CMEs

