CME INDIA Presentation by Dr. S. K. Gupta, Consultant Physician, Delhi.

1. ✅ Indications (Evidence-based)
| Clinical setting | Practical use |
| Type 2 Diabetes Mellitus (T2DM) | Inadequate control on metformin ± other OADs |
| ASCVD / high CV risk | Proven CV benefit with semaglutide class data; subcutaneous semaglutide reduced major adverse cardiovascular events in SUSTAIN-6, and oral semaglutide also showed MACE reduction in SOUL. |
| Obesity / Overweight | BMI ≥30 kg/m² OR BMI ≥27 kg/m² + comorbidity; semaglutide 2.4 mg has proven weight-loss efficacy and cardiovascular benefit in eligible overweight/obesity populations. |
| Cardiorenal benefit | Reduces MACE; kidney benefit shown in FLOW in T2DM + CKD. |
- Particularly useful in:
Insulin resistance
NAFLD / MASLD
PCOS
Cardiometabolic risk clustering
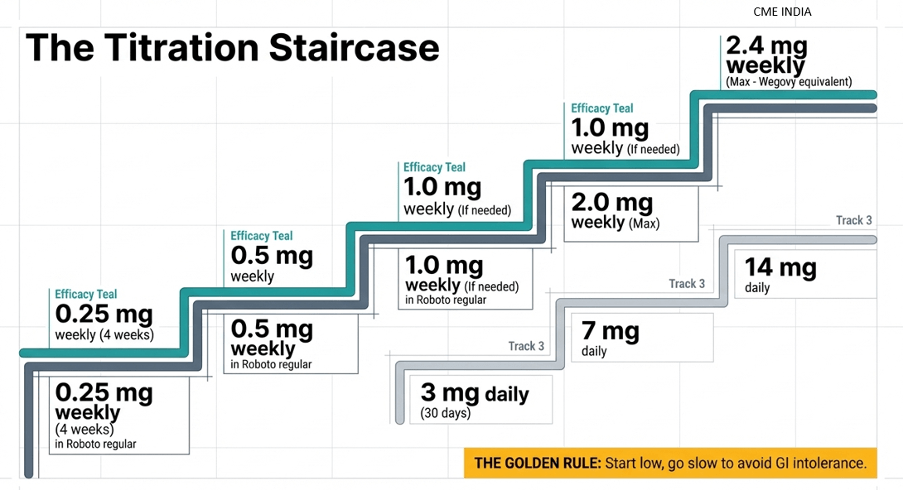
2. Dosage & Titration (Key to tolerability)
A. Subcutaneous Semaglutide
| Situation | Usual titration |
| T2DM (subcutaneous) | Start 0.25 mg once weekly for 4 weeks, then 0.5 mg weekly; if needed increase to 1 mg, then 2 mg. |
| Obesity (subcutaneous semaglutide 2.4 mg) | Escalate gradually to 2.4 mg weekly to improve tolerability. |
B. Oral Semaglutide
Important update: the older 3 mg → 7 mg → 14 mg sequence applied to the earlier oral formulation. The updated U.S. label now also includes an R2 formulation with a different titration schedule
| Oral semaglutide version | Practical note |
| Older formulation | 3 mg daily for 30 days → 7 mg → 14 mg |
| Updated R2 formulation (latest U.S. label) | 1.5 mg daily for 30 days → 4 mg for 30 days → then higher maintenance per response/tolerability. (FDA Access Data) |
👉 Golden rule: Start low, go slow to avoid GI intolerance. This remains clinically correct and is consistent with label-based dose-escalation logic.
3. ⚙️ Mechanism (for clinical insight)
GLP-1 receptor agonist
↓ Appetite (central satiety)
↓ Gastric emptying
↑ Glucose-dependent insulin secretion
↓ Glucagon
This mechanism underlies both glycemic benefit and weight reduction.
4. ⚠️ Common Side Effects
| Very common / dose-dependent | Important clinical concerns |
| Nausea | Pancreatitis warning |
| Vomiting | Gallbladder disease / cholelithiasis / cholecystitis |
| Early satiety | Dehydration and AKI risk, especially if vomiting persists |
| Constipation / diarrhea | Worsening diabetic retinopathy can occur with rapid glucose improvement in some patients |
These adverse effects and warnings are reflected in current prescribing information and major trials.
5. Special Situations & High-Risk Groups
🔴 Immunocompromised patients
No direct immunosuppression.
BUT: risk of reduced intake, malnutrition, and excessive weight loss in fragile patients remains a practical concern. This is more a clinical caution than a formal label contraindication.
\Monitor:
Weight loss speed
Protein intake
Use cautiously in:
Cancer cachexia
Chronic infections such as TB / HIV when nutritional reserve is poor
🔴 Renal disease
Generally no dose adjustment required in CKD, but vomiting/dehydration can precipitate AKI
🔴 Hepatic disease
Generally safe; often useful in obesity / fatty liver settings, though not a liver-specific approved indication.
🔴 Elderly / Frail
Start ultra-low and slow.
Avoid sarcopenia.
Ensure protein intake, mobility, and resistance activity. ADA 2026 emphasizes individualized care in older adults, with attention to nutrition, functional status, and overtreatment avoidance
🔴 Thyroid caution
Avoid in:
Medullary thyroid carcinoma (MTC)
MEN-2 syndrome
This remains a boxed warning / contraindication in current labels.
6. Drug Interactions (Very Important in Practice)
| Interaction issue | Practical implication |
| Delayed gastric emptying | May affect absorption of some oral drugs |
| Sulfonylurea / insulin | Hypoglycemia risk rises; reduce dose when needed |
| Diuretics | Dehydration risk if GI side effects occur |
| NSAIDs | AKI risk may rise in volume depletion |
- Examples commonly discussed in practice:
Levothyroxine
Antibiotics
Oral contraceptives
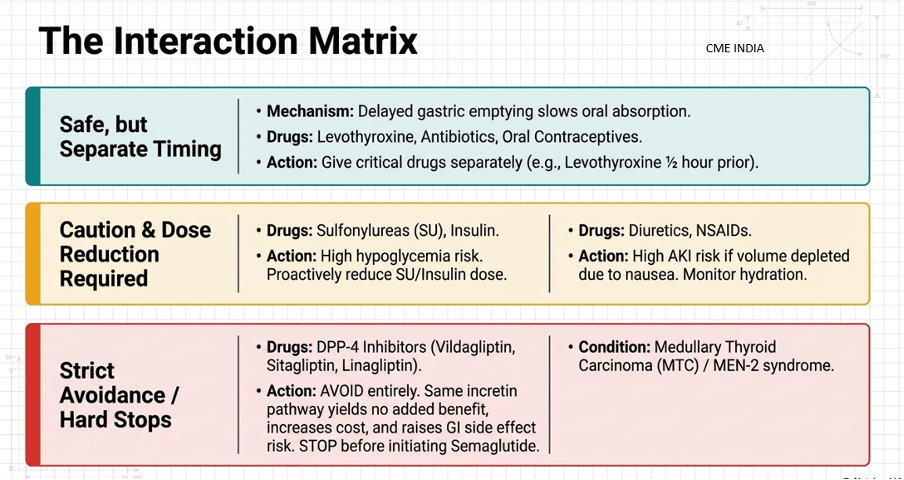
👉 Advice: give critical oral drugs carefully and monitor clinical response when GI slowing is prominent. Current labels do warn that delayed gastric emptying may affect oral medication absorption.
Your sentence “Semaglutide is ~10x more potent than tirzepatide” should be removed from academic or prescribing material because it is not an evidence-based clinical conversion rule. There is no validated dose-conversion ratio between tirzepatide and semaglutide.
Important correction:
“Give levothyroxine 1/2 hour before semaglutide” is not a standard universal rule for weekly injectable semaglutide. For oral semaglutide, timing instructions are formulation-specific and much more important.
7. Monitoring Protocol
| Baseline | Follow-up |
| HbA1c | Weight: monthly |
| Weight, BMI | HbA1c: every 3 months |
| Renal function | Renal function if GI symptoms / dehydration |
| Liver profile | Monitor tolerability and intake |
| Lipid profile | Watch for persistent vomiting or abdominal pain |
Watch for:
Persistent vomiting
Abdominal pain
Visual symptoms
Excessive weight loss
Weakness / falls / sarcopenia signals
This is good practical OPD monitoring and is aligned with real-world use, though exact frequency should be individualized.

8. When to STOP / HOLD
Suspected pancreatitis
Severe GI intolerance
Rapid unexplained weight loss
Severe dehydration
Before major surgery / procedures where delayed gastric emptying is a concern
Practical update: peri-procedural withholding recommendations have evolved over time and can vary by procedure, aspiration risk, and society guidance. A blanket “stop 1 week prior” is often used in practice for weekly dosing, but local anesthesia / endoscopy / anesthesia protocols should be checked individually.
9. Practical Pearls (Indian Context)
- High misuse risk in “cosmetic weight loss clinics”
Counsel patients:
This is not a lifestyle shortcut
Needs diet + exercise
Needs protein preservation
Needs follow-up - Avoid:
BMI <25 cosmetic use without proper indication - Watch:
Lean diabetics common in India
Risk of excessive weight loss
Risk of muscle loss
Risk of frailty in older adults
Most important to know about Semaglutide which no Pharma House tells
Muscle Loss (Sarcopenia) with Semaglutide – Clinical Caution
Why it happens?
Rapid weight loss = loss of both fat + lean mass
Appetite suppression → reduced protein intake
Sedentary patients → muscle catabolism
Studies of semaglutide-associated weight loss show that lean mass does decrease, although total body composition usually still improves because fat mass falls more. The practical concern is greatest in frail, older, sedentary, under-proteinized, or already sarcopenic patients
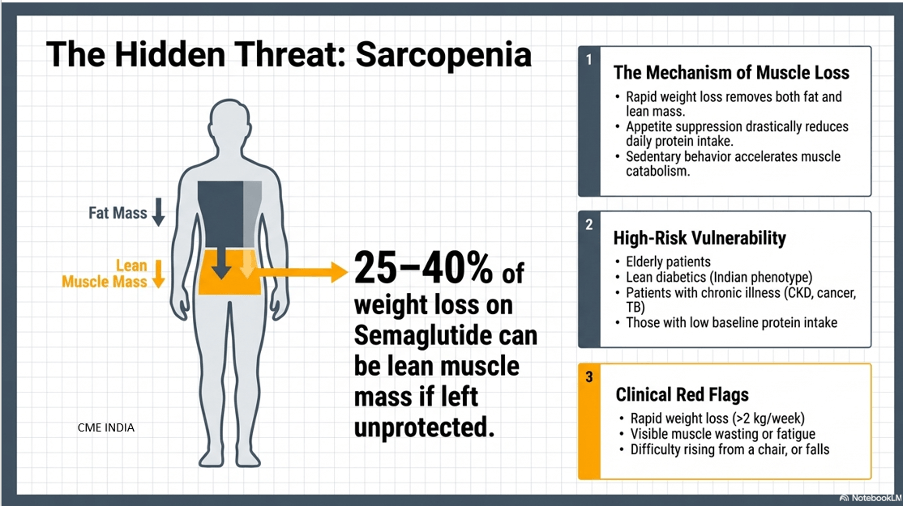
Who is at HIGH RISK?
-Elderly patients
-Lean diabetics (Indian phenotype)
-Chronic illness (CKD, cancer, TB)
-Low baseline protein intake
-Sedentary lifestyle
-Frailty / poor reserve
🛡️ Prevention Strategy (Must prescribe along with drug)
1. Exercise – NON-NEGOTIABLE
Resistance training is most important
3–4 times/week
Even simple:
Chair squats
Wall push-ups
Light weights
Aerobic:
Walking 30 min/day
👉 Without resistance exercise, muscle loss risk rises.
2. Protein Intake
Target usually around 1.0–1.2 g/kg/day, and in selected obesity/weight-loss settings may need more if renal status permits.
Practical Indian advice:
Paneer
Curd
Dal
Chana
Eggs
Protein supplements if intake poor
👉 Divide protein across meals.
3. Calcium & Vitamin D
Helpful where intake is poor, deficiency is likely, or bone risk is present.
Not a semaglutide-specific rule for every patient, but very relevant in older adults and rapid weight-loss settings.
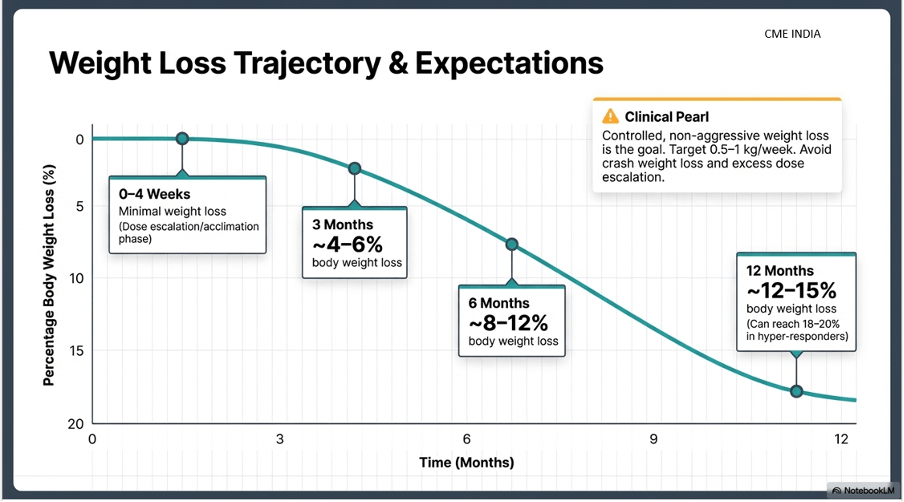
4. Weight Loss Target – Controlled, not aggressive
Ideal:
0.5–1 kg/week in many patients
Avoid:
Crash weight loss
Excess dose escalation
5. Monitoring for Sarcopenia
Clinical signs:
Weakness
Difficulty rising from chair
Fatigue
Falls
Simple OPD tools:
Hand grip strength
Mid-arm circumference
Gait speed
🚨 Red flags – Act immediately
Rapid weight loss (>2 kg/week)
Visible muscle wasting
Fatigue, falls
👉 Action:
Reduce dose
Increase protein
Add resistance work
Reassess treatment goal
Semaglutide: Know before you Login
📌 1. 📉 Expected Weight Loss – “How much, how fast, in whom?”
| Time point | Practical expectation |
| 0–4 weeks | Minimal; dose-escalation phase |
| 3 months | ~4–6% in many responders |
| 6 months | ~8–12% in many responders |
| 12 months | ~12–15% typical in strong responders; some achieve more |
These estimates are broadly consistent with STEP-era evidence and real-world expectations, but they vary by dose, adherence, baseline weight, diabetes status, and tolerability
🔹 Who responds BEST
Obese (BMI >30)
High insulin resistance
Central obesity / fatty liver
Good adherence
Adequate protein intake
No severe GI intolerance
🔹 Who responds POORLY
Lean T2DM
Long-standing diabetes on insulin
Emotional eaters / binge eating
Sedentary lifestyle
👉 Clinical pearl:
If <5% weight loss at 3 months on a meaningful therapeutic dose, reassess adherence, dose, nutrition, activity, and whether this is the right drug for the right patient.
📌 2. ⚠️ Avoid combination with DPP-4 inhibitors
Vildagliptin
Sitagliptin
Linagliptin
❌ Why?
Same incretin pathway
Little added efficacy
More cost
More polypharmacy
👉 Rule in practice: stop DPP-4 inhibitor before starting semaglutide unless there is a very unusual reason not to. This is consistent with diabetes guideline logic.
📌 3. 🔄 Switching: Tirzepatide → Semaglutide
When to switch?
Cost issues
GI intolerance
Availability
Patient preference
Practical approach
No formal washout is usually required in routine practice; many clinicians start the next weekly agent about 1 week after the last dose of the prior weekly incretin, then titrate cautiously.
4. Side Effects Monitoring (Practical OPD Protocol)
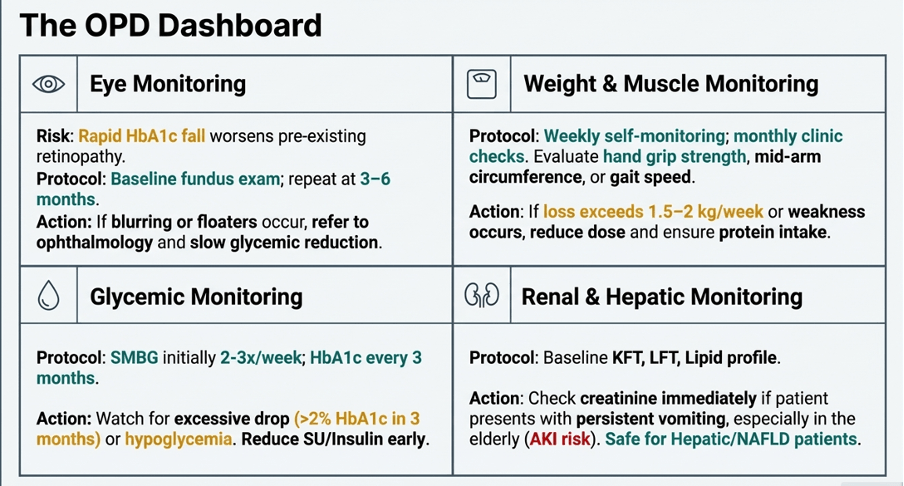
👁️ A. Eye Monitoring (Retinopathy risk)
Why?
Rapid HbA1c fall may transiently worsen diabetic retinopathy in susceptible patients. This signal was noted in SUSTAIN-6 and is also reflected in current labelling.
Who is high-risk?
Pre-existing diabetic retinopathy
Very high HbA1c
Rapid glucose drop
What to do?
| Baseline fundus exam in at-risk patients Repeat at 3–6 months if high-risk |
| 🚨 Red flags: Blurring of vision Floaters Visual change |
| ⚖️ B. Weight Monitoring |
| Weekly self-monitoring Monthly clinic review |
| 🚨 Concern if: 1.5–2 kg/week loss Weakness Sarcopenia features |
| 🍬 C. Sugar Monitoring |
| SMBG initially when clinically needed HbA1c every 3 months |
| 🚨 Watch for: Hypoglycemia if on insulin / SU Excessively rapid glycemic drop |
| 💧 D. Dehydration & Renal Monitoring |
| Check creatinine if: Persistent vomiting Elderly CKD Poor intake |
📌 5. 🚨 When Side Effects Occur – What to Do
GI intolerance
Hold dose escalation
Step back to previous dose
Small frequent meals
Persistent vomiting
Stop drug temporarily
Hydrate
Check renal function
Suspected pancreatitis
Severe epigastric pain → STOP immediately
Check amylase / lipase
Evaluate urgently
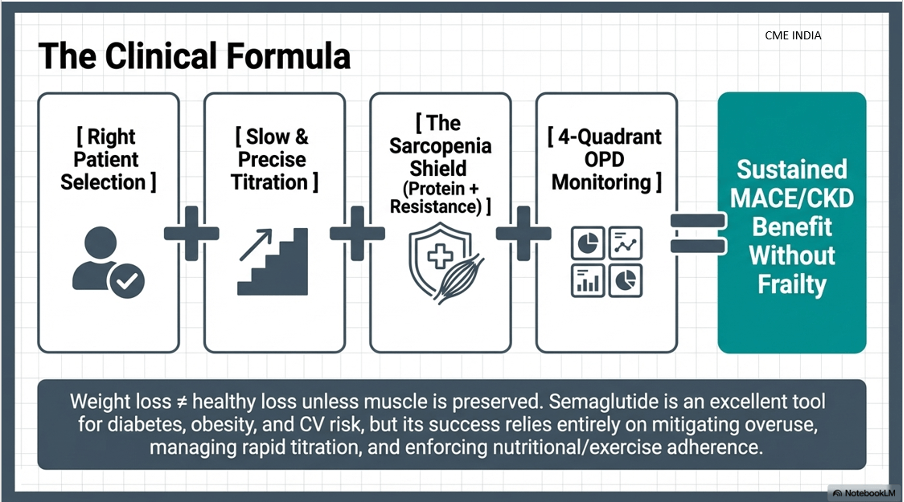
🧠 Final Clinical Takeaways
✔ Expect meaningful weight loss in the right patient, but not every patient loses equally
✔ Avoid DPP-4 inhibitors alongside semaglutide
✔ Switching from tirzepatide to semaglutide should be cautious; there is no validated fixed conversion ratio
✔ Monitor:
👁 Eye
⚖ Weight
🍬 Sugar
💧 Renal function
💪 Muscle preservation
✔ Always co-prescribe:
Exercise
Protein
Monitoring
Counseling
❗ Otherwise risk rises for:
Sarcopenia
Frailty
Falls
Discontinuation
Poor long-term outcomes
References:
- Marso SP, Bain SC, Consoli A, et al. Semaglutide and Cardiovascular Outcomes in Patients with Type 2 Diabetes. N Engl J Med. 2016;375:1834-1844. doi:10.1056/NEJMoa1607141.
- Wilding JPH, Batterham RL, Calanna S, et al. Once-Weekly Semaglutide in Adults with Overweight or Obesity. N Engl J Med. 2021;384:989-1002. doi:10.1056/NEJMoa2032183.
- Lincoff AM, Brown-Frandsen K, Colhoun HM, et al. Semaglutide and Cardiovascular Outcomes in Obesity without Diabetes. N Engl J Med. 2023;389:2221-2232. doi:10.1056/NEJMoa2307563.
- Perkovic V, Tuttle KR, Rossing P, et al. Effects of Semaglutide on Chronic Kidney Disease in Patients with Type 2 Diabetes. N Engl J Med. 2024;390:1095-1108. doi:10.1056/NEJMoa2403347.
- McGuire DK, Deanfield J, Husain M, et al. Oral Semaglutide and Cardiovascular Outcomes in High-Risk Type 2 Diabetes. N Engl J Med. 2025. doi:10.1056/NEJMoa2501006.

Discover CME INDIA

- Explore CME INDIA Repository
- Examine CME INDIA Case Study
- Read History Today in Medicine
- Register for Future CMEs

AI generated contents should be proof-red before uploading.. “Semaglutide is 10x more potent than Tirzepatide!!”
We highly appreciate your comment.
We will do needful