CME INDIA Presentation by Dr. Prakash, M.D (GM)., D. Diab. FICP, Professor & Head, Department of Diabetology, Government Mohan Kumara Mangalam Medical College; Senior Consultant Diabetologist, Salem Diabetes Foundation, Salem, Tamil Nadu.
Based on a presentation at APICON 2024, New Delhi on 23/2/2024.
CGMS plays a crucial role
- Effective management of glycemia (as demonstrated by DCCT and UKPDS) and its associated complications.
- The transition from urine sugar testing in the past to contemporary Continuous Glucose Monitoring Systems (CGMS).
- Introduced in the late 1990s, CGMS provides continuous subcutaneous monitoring of interstitial glucose levels.
- CGMS stands out as a valuable tool in evaluating glucose fluctuations, offering accuracy, convenience, and benefiting from ongoing software advancements.
- While HbA1C continues to serve as a primary surrogate marker for glycemic control, CGMS plays a crucial role in informing therapeutic decisions.
SMBG VS CGMS
- Self-Monitoring of Blood Glucose (SMBG) captures blood sugar levels at a single moment, unable to identify nocturnal or asymptomatic hypoglycemia and glycemic fluctuations.
- Continuous Glucose Monitoring Systems (CGMS) track the rate of blood sugar level changes and highlight glucose trends.
- CGMS furnishes insights into glucose level excursions within a day or across days.
- CGMS reveals glucose patterns during overnight fasting, postprandial states of 2-4 hours, and the timing of insulin and medication.
- By reducing the risk of hypoglycemia and maintaining glycemic stability, CGMS serves to minimize glycemic variability.
CGMS versus HbA1c
- HbA1C fails to capture the nuances of inter and intra glucose excursions arising from hypoglycemia or postprandial states.
- CGMS technology tracks day-to-day glycemic excursions and glucose variability, aiding in the pursuit of glycemic goals.
- CGMS enhances treatment decisions to attain and maintain glycemic targets.
Mechanism:
- CGMS is continuous subcutaneous quantification of interstitial glucose levels.
- It consists of a small disposable sensor which is inserted into the subcutaneous tissue, a transmitter and receiver.
- The sensor inserted may be either transcutaneous or implantable.
- The transmitter attached to the sensor wirelessly transmits glucose values at 1 min and 5 min intervals to a reader, an App or an Automated Insulin delivery device.
- The transcutaneous system has sensors of wear time from 6 days to 14 days after which a new sensor should be inserted.
- Implantable systems can transmit glucose levels for up to 180 days before replacement.
- CGMS sensors that are available now are mostly factory calibrated.
- The receiver provides with trends in glucose levels and alert the user as well when glucose levels exceed or fall below the predetermined thresholds.
Types of CGM
| Type of CGM | Description |
| rt CGM (Real time CGM) | Measures and display glucose levels continuously |
| is CGM – with or without alarm (Intermittently scanned CGM) | Measures glucose levels continuously but require scanning for visualization and storage of glucose values. |
| Professional CGM | These devices are clinic based and not owned by the person with Diabetes Mellitus. The device is worn for a desirable period of time (7-14) days. Data may be blinded or visible to the person who wear the device. |
| I cgm (Integrated CGM) | A higher standard set by FDA where the device can be integrated with other digitally connected devices. |
Real time CGM systems are also classified as adjunctive where user has to perform self-glucose monitoring and nonadjunctive where devices are authorized by regulators to replace SMBG
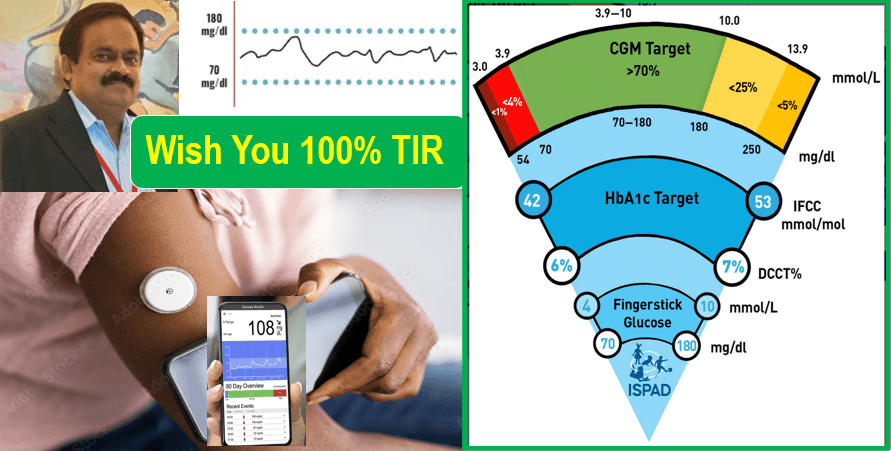
Time in Range (TIR)
- TIR – The time spent by the individual within the target range of glucose.
- Key metric currently used in various current clinical trials.
- DCCT data provided intensive treatment group had substantially higher TIR compared to conventional therapy.
- TIR measurement helps in evaluation and comparing intervention of various glucose lowering agents
- The Time in Range of 70-180 mgs was demonstrated as target from quarterly seven-point blood glucose testing of DCCT data.
- The TIR had a strong association with the risk of microalbuminuria, retinopathy development and its progression.
- The goal of TIR of 70% align with HbA1C of 7%.
- Time below range (TBR <70 mgs and <54 mgs).
- Time above range (TAR >180 mgs) are useful parameters for adjustment of insulin dose and modification of treatment plan.
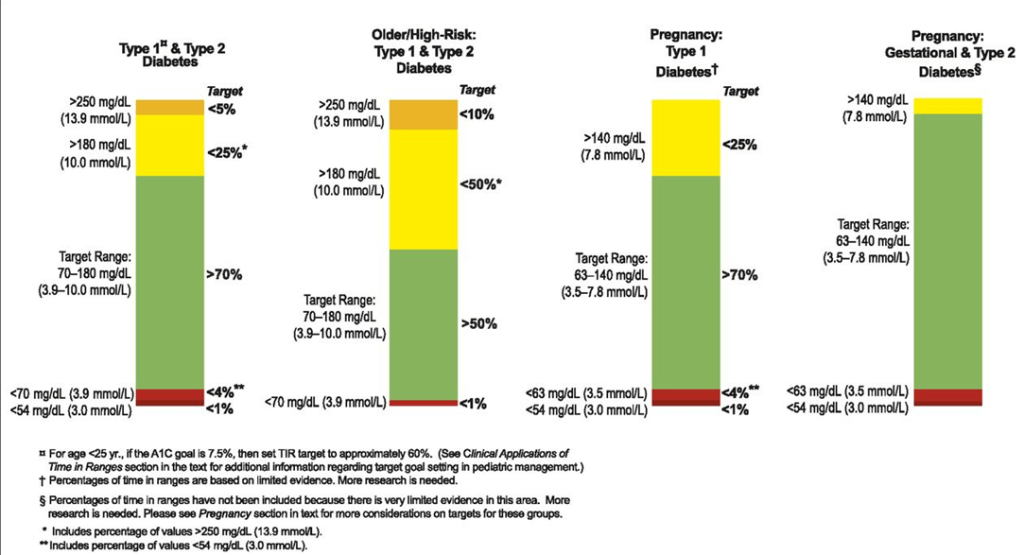
Courtesy: Tadej Battelino, Thomas Danne, Richard M. Bergenstal, Stephanie A. Amiel,et al. Clinical Targets for Continuous Glucose Monitoring Data Interpretation: Recommendations From the International Consensus on Time in Range. Diabetes Care 1 August 2019; 42 (8): 1593–1603.


Side Effects:
- Contact dermatitis (both irritant and allergic)
Substances Interfering with CGM Devices

Quantifying CGM Accuracy:
- Mean Absolute Relative Difference (MARD)
- Assess the accuracy of CGM devices.
- Averaging the absolute values of the relative differences between a CGM measured and the corresponding simultaneous value obtained by the reference system.
- Ranges between 8% and 14% for the currently available CGM devices.
Indications:
| Type 1 Diabetes Mellitus for achieving glycemic targets. |
| Diabetes Mellitus and Pregnancy to maintain strict control. |
| Type 2 DM, who are on multiple doses on insulin. |
| Diabetes Mellitus having Hypoglycemia unawareness |
| Diabetes Mellitus on insulin pump therapy |
| Pre-Diabetes or Diabetes mellitus who are willing to change their lifestyle behavior and drugs based on CGM readings |
Merits:
- Provides continuous reading of blood glucose every 1-5 minutes.
- Data can be downloaded.
- Glycemic patterns can be analyzed in detail.
- Treatment can be modified based on the pattern of glycemic excursions during sleep, exercise, before and after food, stress and sexual activity.
- The latest generation devices are factory calibrated (No fingers prick test)
Demerits:
- Expensive
- Time consuming
- Expertise is required
- Physiologic lag time between change in glucose level in the blood and interstitium
- Poor assessment of low glucose
- Sensor displacement
- No long-term outcomes data
Some Case Vignettes:
- The prevailing agreement emphasizes the importance of a comprehensible CGM report that is accessible to both healthcare professionals and individuals with diabetes. While certain terms, such as glucose variability, might be less familiar to many people with diabetes, the consensus values a concise, single-page report.
- This report can be readily reviewed and filed in the electronic medical record by the medical team and serves as a shared decision-making tool with individuals managing diabetes.
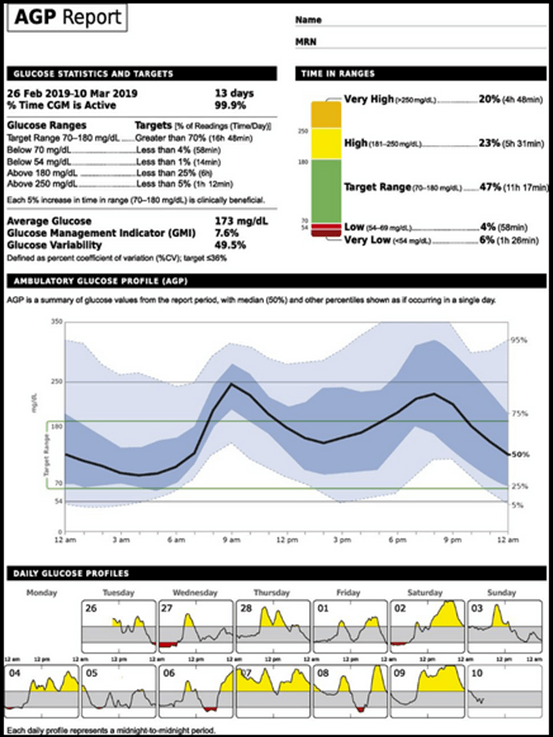
Courtesy: Tadej Battelino, Thomas Danne, Richard M. Bergenstal, Stephanie A. Amiel,et al. Clinical Targets for Continuous Glucose Monitoring Data Interpretation: Recommendations from the International Consensus on Time in Range. Diabetes Care 1 August 2019; 42 (8): 1593–1603.
Case 1: CGM Report of Person with Type 1 Diabetes Mellitus

Case 2: CGM Report of Person with Pre-Gestational Diabetes Mellitus

Case 3: CGM Report of Person with Type 2 Diabetes Mellitus on Insulin therapy

Quick Takeaways
| Comprehensive Diabetes care |
| Improves glycated hemoglobin (HbA1c) |
| Reduces glycemic variability |
| Reduces risk of hypoglycemia |
| Improves quality of life |
| Reduces the number of capillary tests (BGM) |
CME INDIA Learning Points
(Based on Reference 2)
- There have been established guidelines and consensus recommendations regarding the incorporation of CGM-based metrics into routine diabetes care.
- In contrast to traditional glucose metrics like HbA1c, CGM data reports provide supplementary measures of glucose management, including Time in Range (TIR) and glycemic variability.
- TIR, representing the duration spent above, below, or within the glucose target range, stands out as a potent metric for capturing the dynamic fluctuations in blood glucose levels.
- Optimizing diabetes management in the Indian population requires customized CGM metrics, with a specific focus on TIR, differing from the approach in Western populations.
General Indian Recommendations:
- Standard of Care (SOC): In managing glucose for individuals with diabetes, considering Time in Range (TIR) provided by CGM alongside HbA1c is recommended as the new standard of care (SOC). Additionally, other glucose metrics like fasting plasma glucose (FPG) and postprandial glucose (PPG) can be considered. It is essential to be mindful of the limitations of CGM, as it relies on interstitial fluid measurements.
- Increased Awareness of TIR: Both physicians and patients should be more aware of Time in Range (TIR) as a crucial metric in glucose management.
- Applicability in Pandemic Situations: Except for the ICU setting, TIR is strongly recommended in various pandemic scenarios, including homecare, post-hospitalization, quarantine, and in-hospital care.
- Discordance Considerations: Recognize the potential discordance between CGM and glucometer readings of glucose levels. However, recent advancements in CGM devices, such as the FreeStyle Libre system, have reduced such discordances.
(These overall recommendations aim to integrate TIR from CGM into routine diabetes care, emphasizing its significance in various healthcare settings and pandemic situations while acknowledging and addressing potential limitations)
Recommendations for Glucose Management in Pregnancy:
- Women with Type 1 Diabetes (T1DM) planning to conceive:
- Target TIR (Time in Range): ≥ 70% within the range of 70–140 mg/dL.
- Women with Type 2 Diabetes (T2DM), first-time detected hyperglycemic women during pregnancy, or Gestational Diabetes Mellitus (GDM):
- Target TIR: ≥ 90% within the range of 70–140 mg/dL.
These guidelines provide specific TIR targets for glucose management tailored to different scenarios during pregnancy. It underscores the importance of maintaining optimal glucose levels to ensure the well-being of both the mother and the developing fetus, with variations based on the type of diabetes and the stage of pregnancy.
References:
- Battelino, Tadej, et al. “Clinical targets for continuous glucose monitoring data interpretation: recommendations from the international consensus on time in range.” Diabetes care 42.8 (2019): 1593-1603.
- Mohan, V., Joshi, S., Mithal, A. et al. Expert Consensus Recommendations on Time in Range for Monitoring Glucose Levels in People with Diabetes: An Indian Perspective. Diabetes Ther 14, 237–249 (2023). https://doi.org/10.1007/s13300-022-01355-4.
- Martin de Bock, Ethel Codner, Maria E. Craig, Tony Huynh, David M. Maahs, Farid H. Mahmud, Loredana Marcovecchio, Linda A. DiMeglio ISPAD Clinical Practice Consensus Guidelines 2022: Glycemic targets and glucose monitoring for children, adolescents, and young people with diabetes. 20 December 2022. https://doi.org/10.1111/pedi.13455.

Discover CME INDIA

- Explore CME INDIA Repository
- Examine CME INDIA Case Study
- Read History Today in Medicine
- Register for Future CMEs


Excellent article Sir! This is the need of the hour in all Uncontrolled diabetics. In the detail of CGM devices, the ones working thru sensor & others which are Mobile App based could be delineated too.